- Resources Home 🏠
- Try SciSpace Copilot
- Search research papers
- Add Copilot Extension
- Try AI Detector
- Try Paraphraser
- Try Citation Generator
- April Papers
- June Papers
- July Papers


The Craft of Writing a Strong Hypothesis

Table of Contents
Writing a hypothesis is one of the essential elements of a scientific research paper. It needs to be to the point, clearly communicating what your research is trying to accomplish. A blurry, drawn-out, or complexly-structured hypothesis can confuse your readers. Or worse, the editor and peer reviewers.
A captivating hypothesis is not too intricate. This blog will take you through the process so that, by the end of it, you have a better idea of how to convey your research paper's intent in just one sentence.
What is a Hypothesis?
The first step in your scientific endeavor, a hypothesis, is a strong, concise statement that forms the basis of your research. It is not the same as a thesis statement , which is a brief summary of your research paper .
The sole purpose of a hypothesis is to predict your paper's findings, data, and conclusion. It comes from a place of curiosity and intuition . When you write a hypothesis, you're essentially making an educated guess based on scientific prejudices and evidence, which is further proven or disproven through the scientific method.
The reason for undertaking research is to observe a specific phenomenon. A hypothesis, therefore, lays out what the said phenomenon is. And it does so through two variables, an independent and dependent variable.
The independent variable is the cause behind the observation, while the dependent variable is the effect of the cause. A good example of this is “mixing red and blue forms purple.” In this hypothesis, mixing red and blue is the independent variable as you're combining the two colors at your own will. The formation of purple is the dependent variable as, in this case, it is conditional to the independent variable.
Different Types of Hypotheses

Types of hypotheses
Some would stand by the notion that there are only two types of hypotheses: a Null hypothesis and an Alternative hypothesis. While that may have some truth to it, it would be better to fully distinguish the most common forms as these terms come up so often, which might leave you out of context.
Apart from Null and Alternative, there are Complex, Simple, Directional, Non-Directional, Statistical, and Associative and casual hypotheses. They don't necessarily have to be exclusive, as one hypothesis can tick many boxes, but knowing the distinctions between them will make it easier for you to construct your own.
1. Null hypothesis
A null hypothesis proposes no relationship between two variables. Denoted by H 0 , it is a negative statement like “Attending physiotherapy sessions does not affect athletes' on-field performance.” Here, the author claims physiotherapy sessions have no effect on on-field performances. Even if there is, it's only a coincidence.
2. Alternative hypothesis
Considered to be the opposite of a null hypothesis, an alternative hypothesis is donated as H1 or Ha. It explicitly states that the dependent variable affects the independent variable. A good alternative hypothesis example is “Attending physiotherapy sessions improves athletes' on-field performance.” or “Water evaporates at 100 °C. ” The alternative hypothesis further branches into directional and non-directional.
- Directional hypothesis: A hypothesis that states the result would be either positive or negative is called directional hypothesis. It accompanies H1 with either the ‘<' or ‘>' sign.
- Non-directional hypothesis: A non-directional hypothesis only claims an effect on the dependent variable. It does not clarify whether the result would be positive or negative. The sign for a non-directional hypothesis is ‘≠.'
3. Simple hypothesis
A simple hypothesis is a statement made to reflect the relation between exactly two variables. One independent and one dependent. Consider the example, “Smoking is a prominent cause of lung cancer." The dependent variable, lung cancer, is dependent on the independent variable, smoking.
4. Complex hypothesis
In contrast to a simple hypothesis, a complex hypothesis implies the relationship between multiple independent and dependent variables. For instance, “Individuals who eat more fruits tend to have higher immunity, lesser cholesterol, and high metabolism.” The independent variable is eating more fruits, while the dependent variables are higher immunity, lesser cholesterol, and high metabolism.
5. Associative and casual hypothesis
Associative and casual hypotheses don't exhibit how many variables there will be. They define the relationship between the variables. In an associative hypothesis, changing any one variable, dependent or independent, affects others. In a casual hypothesis, the independent variable directly affects the dependent.
6. Empirical hypothesis
Also referred to as the working hypothesis, an empirical hypothesis claims a theory's validation via experiments and observation. This way, the statement appears justifiable and different from a wild guess.
Say, the hypothesis is “Women who take iron tablets face a lesser risk of anemia than those who take vitamin B12.” This is an example of an empirical hypothesis where the researcher the statement after assessing a group of women who take iron tablets and charting the findings.
7. Statistical hypothesis
The point of a statistical hypothesis is to test an already existing hypothesis by studying a population sample. Hypothesis like “44% of the Indian population belong in the age group of 22-27.” leverage evidence to prove or disprove a particular statement.
Characteristics of a Good Hypothesis
Writing a hypothesis is essential as it can make or break your research for you. That includes your chances of getting published in a journal. So when you're designing one, keep an eye out for these pointers:
- A research hypothesis has to be simple yet clear to look justifiable enough.
- It has to be testable — your research would be rendered pointless if too far-fetched into reality or limited by technology.
- It has to be precise about the results —what you are trying to do and achieve through it should come out in your hypothesis.
- A research hypothesis should be self-explanatory, leaving no doubt in the reader's mind.
- If you are developing a relational hypothesis, you need to include the variables and establish an appropriate relationship among them.
- A hypothesis must keep and reflect the scope for further investigations and experiments.
Separating a Hypothesis from a Prediction
Outside of academia, hypothesis and prediction are often used interchangeably. In research writing, this is not only confusing but also incorrect. And although a hypothesis and prediction are guesses at their core, there are many differences between them.
A hypothesis is an educated guess or even a testable prediction validated through research. It aims to analyze the gathered evidence and facts to define a relationship between variables and put forth a logical explanation behind the nature of events.
Predictions are assumptions or expected outcomes made without any backing evidence. They are more fictionally inclined regardless of where they originate from.
For this reason, a hypothesis holds much more weight than a prediction. It sticks to the scientific method rather than pure guesswork. "Planets revolve around the Sun." is an example of a hypothesis as it is previous knowledge and observed trends. Additionally, we can test it through the scientific method.
Whereas "COVID-19 will be eradicated by 2030." is a prediction. Even though it results from past trends, we can't prove or disprove it. So, the only way this gets validated is to wait and watch if COVID-19 cases end by 2030.
Finally, How to Write a Hypothesis

Quick tips on writing a hypothesis
1. Be clear about your research question
A hypothesis should instantly address the research question or the problem statement. To do so, you need to ask a question. Understand the constraints of your undertaken research topic and then formulate a simple and topic-centric problem. Only after that can you develop a hypothesis and further test for evidence.
2. Carry out a recce
Once you have your research's foundation laid out, it would be best to conduct preliminary research. Go through previous theories, academic papers, data, and experiments before you start curating your research hypothesis. It will give you an idea of your hypothesis's viability or originality.
Making use of references from relevant research papers helps draft a good research hypothesis. SciSpace Discover offers a repository of over 270 million research papers to browse through and gain a deeper understanding of related studies on a particular topic. Additionally, you can use SciSpace Copilot , your AI research assistant, for reading any lengthy research paper and getting a more summarized context of it. A hypothesis can be formed after evaluating many such summarized research papers. Copilot also offers explanations for theories and equations, explains paper in simplified version, allows you to highlight any text in the paper or clip math equations and tables and provides a deeper, clear understanding of what is being said. This can improve the hypothesis by helping you identify potential research gaps.
3. Create a 3-dimensional hypothesis
Variables are an essential part of any reasonable hypothesis. So, identify your independent and dependent variable(s) and form a correlation between them. The ideal way to do this is to write the hypothetical assumption in the ‘if-then' form. If you use this form, make sure that you state the predefined relationship between the variables.
In another way, you can choose to present your hypothesis as a comparison between two variables. Here, you must specify the difference you expect to observe in the results.
4. Write the first draft
Now that everything is in place, it's time to write your hypothesis. For starters, create the first draft. In this version, write what you expect to find from your research.
Clearly separate your independent and dependent variables and the link between them. Don't fixate on syntax at this stage. The goal is to ensure your hypothesis addresses the issue.
5. Proof your hypothesis
After preparing the first draft of your hypothesis, you need to inspect it thoroughly. It should tick all the boxes, like being concise, straightforward, relevant, and accurate. Your final hypothesis has to be well-structured as well.
Research projects are an exciting and crucial part of being a scholar. And once you have your research question, you need a great hypothesis to begin conducting research. Thus, knowing how to write a hypothesis is very important.
Now that you have a firmer grasp on what a good hypothesis constitutes, the different kinds there are, and what process to follow, you will find it much easier to write your hypothesis, which ultimately helps your research.
Now it's easier than ever to streamline your research workflow with SciSpace Discover . Its integrated, comprehensive end-to-end platform for research allows scholars to easily discover, write and publish their research and fosters collaboration.
It includes everything you need, including a repository of over 270 million research papers across disciplines, SEO-optimized summaries and public profiles to show your expertise and experience.
If you found these tips on writing a research hypothesis useful, head over to our blog on Statistical Hypothesis Testing to learn about the top researchers, papers, and institutions in this domain.
Frequently Asked Questions (FAQs)
1. what is the definition of hypothesis.
According to the Oxford dictionary, a hypothesis is defined as “An idea or explanation of something that is based on a few known facts, but that has not yet been proved to be true or correct”.
2. What is an example of hypothesis?
The hypothesis is a statement that proposes a relationship between two or more variables. An example: "If we increase the number of new users who join our platform by 25%, then we will see an increase in revenue."
3. What is an example of null hypothesis?
A null hypothesis is a statement that there is no relationship between two variables. The null hypothesis is written as H0. The null hypothesis states that there is no effect. For example, if you're studying whether or not a particular type of exercise increases strength, your null hypothesis will be "there is no difference in strength between people who exercise and people who don't."
4. What are the types of research?
• Fundamental research
• Applied research
• Qualitative research
• Quantitative research
• Mixed research
• Exploratory research
• Longitudinal research
• Cross-sectional research
• Field research
• Laboratory research
• Fixed research
• Flexible research
• Action research
• Policy research
• Classification research
• Comparative research
• Causal research
• Inductive research
• Deductive research
5. How to write a hypothesis?
• Your hypothesis should be able to predict the relationship and outcome.
• Avoid wordiness by keeping it simple and brief.
• Your hypothesis should contain observable and testable outcomes.
• Your hypothesis should be relevant to the research question.
6. What are the 2 types of hypothesis?
• Null hypotheses are used to test the claim that "there is no difference between two groups of data".
• Alternative hypotheses test the claim that "there is a difference between two data groups".
7. Difference between research question and research hypothesis?
A research question is a broad, open-ended question you will try to answer through your research. A hypothesis is a statement based on prior research or theory that you expect to be true due to your study. Example - Research question: What are the factors that influence the adoption of the new technology? Research hypothesis: There is a positive relationship between age, education and income level with the adoption of the new technology.
8. What is plural for hypothesis?
The plural of hypothesis is hypotheses. Here's an example of how it would be used in a statement, "Numerous well-considered hypotheses are presented in this part, and they are supported by tables and figures that are well-illustrated."
9. What is the red queen hypothesis?
The red queen hypothesis in evolutionary biology states that species must constantly evolve to avoid extinction because if they don't, they will be outcompeted by other species that are evolving. Leigh Van Valen first proposed it in 1973; since then, it has been tested and substantiated many times.
10. Who is known as the father of null hypothesis?
The father of the null hypothesis is Sir Ronald Fisher. He published a paper in 1925 that introduced the concept of null hypothesis testing, and he was also the first to use the term itself.
11. When to reject null hypothesis?
You need to find a significant difference between your two populations to reject the null hypothesis. You can determine that by running statistical tests such as an independent sample t-test or a dependent sample t-test. You should reject the null hypothesis if the p-value is less than 0.05.
You might also like

Consensus GPT vs. SciSpace GPT: Choose the Best GPT for Research

Literature Review and Theoretical Framework: Understanding the Differences

Types of Essays in Academic Writing - Quick Guide (2024)
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- J Korean Med Sci
- v.37(16); 2022 Apr 25

A Practical Guide to Writing Quantitative and Qualitative Research Questions and Hypotheses in Scholarly Articles
Edward barroga.
1 Department of General Education, Graduate School of Nursing Science, St. Luke’s International University, Tokyo, Japan.
Glafera Janet Matanguihan
2 Department of Biological Sciences, Messiah University, Mechanicsburg, PA, USA.
The development of research questions and the subsequent hypotheses are prerequisites to defining the main research purpose and specific objectives of a study. Consequently, these objectives determine the study design and research outcome. The development of research questions is a process based on knowledge of current trends, cutting-edge studies, and technological advances in the research field. Excellent research questions are focused and require a comprehensive literature search and in-depth understanding of the problem being investigated. Initially, research questions may be written as descriptive questions which could be developed into inferential questions. These questions must be specific and concise to provide a clear foundation for developing hypotheses. Hypotheses are more formal predictions about the research outcomes. These specify the possible results that may or may not be expected regarding the relationship between groups. Thus, research questions and hypotheses clarify the main purpose and specific objectives of the study, which in turn dictate the design of the study, its direction, and outcome. Studies developed from good research questions and hypotheses will have trustworthy outcomes with wide-ranging social and health implications.
INTRODUCTION
Scientific research is usually initiated by posing evidenced-based research questions which are then explicitly restated as hypotheses. 1 , 2 The hypotheses provide directions to guide the study, solutions, explanations, and expected results. 3 , 4 Both research questions and hypotheses are essentially formulated based on conventional theories and real-world processes, which allow the inception of novel studies and the ethical testing of ideas. 5 , 6
It is crucial to have knowledge of both quantitative and qualitative research 2 as both types of research involve writing research questions and hypotheses. 7 However, these crucial elements of research are sometimes overlooked; if not overlooked, then framed without the forethought and meticulous attention it needs. Planning and careful consideration are needed when developing quantitative or qualitative research, particularly when conceptualizing research questions and hypotheses. 4
There is a continuing need to support researchers in the creation of innovative research questions and hypotheses, as well as for journal articles that carefully review these elements. 1 When research questions and hypotheses are not carefully thought of, unethical studies and poor outcomes usually ensue. Carefully formulated research questions and hypotheses define well-founded objectives, which in turn determine the appropriate design, course, and outcome of the study. This article then aims to discuss in detail the various aspects of crafting research questions and hypotheses, with the goal of guiding researchers as they develop their own. Examples from the authors and peer-reviewed scientific articles in the healthcare field are provided to illustrate key points.
DEFINITIONS AND RELATIONSHIP OF RESEARCH QUESTIONS AND HYPOTHESES
A research question is what a study aims to answer after data analysis and interpretation. The answer is written in length in the discussion section of the paper. Thus, the research question gives a preview of the different parts and variables of the study meant to address the problem posed in the research question. 1 An excellent research question clarifies the research writing while facilitating understanding of the research topic, objective, scope, and limitations of the study. 5
On the other hand, a research hypothesis is an educated statement of an expected outcome. This statement is based on background research and current knowledge. 8 , 9 The research hypothesis makes a specific prediction about a new phenomenon 10 or a formal statement on the expected relationship between an independent variable and a dependent variable. 3 , 11 It provides a tentative answer to the research question to be tested or explored. 4
Hypotheses employ reasoning to predict a theory-based outcome. 10 These can also be developed from theories by focusing on components of theories that have not yet been observed. 10 The validity of hypotheses is often based on the testability of the prediction made in a reproducible experiment. 8
Conversely, hypotheses can also be rephrased as research questions. Several hypotheses based on existing theories and knowledge may be needed to answer a research question. Developing ethical research questions and hypotheses creates a research design that has logical relationships among variables. These relationships serve as a solid foundation for the conduct of the study. 4 , 11 Haphazardly constructed research questions can result in poorly formulated hypotheses and improper study designs, leading to unreliable results. Thus, the formulations of relevant research questions and verifiable hypotheses are crucial when beginning research. 12
CHARACTERISTICS OF GOOD RESEARCH QUESTIONS AND HYPOTHESES
Excellent research questions are specific and focused. These integrate collective data and observations to confirm or refute the subsequent hypotheses. Well-constructed hypotheses are based on previous reports and verify the research context. These are realistic, in-depth, sufficiently complex, and reproducible. More importantly, these hypotheses can be addressed and tested. 13
There are several characteristics of well-developed hypotheses. Good hypotheses are 1) empirically testable 7 , 10 , 11 , 13 ; 2) backed by preliminary evidence 9 ; 3) testable by ethical research 7 , 9 ; 4) based on original ideas 9 ; 5) have evidenced-based logical reasoning 10 ; and 6) can be predicted. 11 Good hypotheses can infer ethical and positive implications, indicating the presence of a relationship or effect relevant to the research theme. 7 , 11 These are initially developed from a general theory and branch into specific hypotheses by deductive reasoning. In the absence of a theory to base the hypotheses, inductive reasoning based on specific observations or findings form more general hypotheses. 10
TYPES OF RESEARCH QUESTIONS AND HYPOTHESES
Research questions and hypotheses are developed according to the type of research, which can be broadly classified into quantitative and qualitative research. We provide a summary of the types of research questions and hypotheses under quantitative and qualitative research categories in Table 1 .
| Quantitative research questions | Quantitative research hypotheses |
|---|---|
| Descriptive research questions | Simple hypothesis |
| Comparative research questions | Complex hypothesis |
| Relationship research questions | Directional hypothesis |
| Non-directional hypothesis | |
| Associative hypothesis | |
| Causal hypothesis | |
| Null hypothesis | |
| Alternative hypothesis | |
| Working hypothesis | |
| Statistical hypothesis | |
| Logical hypothesis | |
| Hypothesis-testing | |
| Qualitative research questions | Qualitative research hypotheses |
| Contextual research questions | Hypothesis-generating |
| Descriptive research questions | |
| Evaluation research questions | |
| Explanatory research questions | |
| Exploratory research questions | |
| Generative research questions | |
| Ideological research questions | |
| Ethnographic research questions | |
| Phenomenological research questions | |
| Grounded theory questions | |
| Qualitative case study questions |
Research questions in quantitative research
In quantitative research, research questions inquire about the relationships among variables being investigated and are usually framed at the start of the study. These are precise and typically linked to the subject population, dependent and independent variables, and research design. 1 Research questions may also attempt to describe the behavior of a population in relation to one or more variables, or describe the characteristics of variables to be measured ( descriptive research questions ). 1 , 5 , 14 These questions may also aim to discover differences between groups within the context of an outcome variable ( comparative research questions ), 1 , 5 , 14 or elucidate trends and interactions among variables ( relationship research questions ). 1 , 5 We provide examples of descriptive, comparative, and relationship research questions in quantitative research in Table 2 .
| Quantitative research questions | |
|---|---|
| Descriptive research question | |
| - Measures responses of subjects to variables | |
| - Presents variables to measure, analyze, or assess | |
| What is the proportion of resident doctors in the hospital who have mastered ultrasonography (response of subjects to a variable) as a diagnostic technique in their clinical training? | |
| Comparative research question | |
| - Clarifies difference between one group with outcome variable and another group without outcome variable | |
| Is there a difference in the reduction of lung metastasis in osteosarcoma patients who received the vitamin D adjunctive therapy (group with outcome variable) compared with osteosarcoma patients who did not receive the vitamin D adjunctive therapy (group without outcome variable)? | |
| - Compares the effects of variables | |
| How does the vitamin D analogue 22-Oxacalcitriol (variable 1) mimic the antiproliferative activity of 1,25-Dihydroxyvitamin D (variable 2) in osteosarcoma cells? | |
| Relationship research question | |
| - Defines trends, association, relationships, or interactions between dependent variable and independent variable | |
| Is there a relationship between the number of medical student suicide (dependent variable) and the level of medical student stress (independent variable) in Japan during the first wave of the COVID-19 pandemic? | |
Hypotheses in quantitative research
In quantitative research, hypotheses predict the expected relationships among variables. 15 Relationships among variables that can be predicted include 1) between a single dependent variable and a single independent variable ( simple hypothesis ) or 2) between two or more independent and dependent variables ( complex hypothesis ). 4 , 11 Hypotheses may also specify the expected direction to be followed and imply an intellectual commitment to a particular outcome ( directional hypothesis ) 4 . On the other hand, hypotheses may not predict the exact direction and are used in the absence of a theory, or when findings contradict previous studies ( non-directional hypothesis ). 4 In addition, hypotheses can 1) define interdependency between variables ( associative hypothesis ), 4 2) propose an effect on the dependent variable from manipulation of the independent variable ( causal hypothesis ), 4 3) state a negative relationship between two variables ( null hypothesis ), 4 , 11 , 15 4) replace the working hypothesis if rejected ( alternative hypothesis ), 15 explain the relationship of phenomena to possibly generate a theory ( working hypothesis ), 11 5) involve quantifiable variables that can be tested statistically ( statistical hypothesis ), 11 6) or express a relationship whose interlinks can be verified logically ( logical hypothesis ). 11 We provide examples of simple, complex, directional, non-directional, associative, causal, null, alternative, working, statistical, and logical hypotheses in quantitative research, as well as the definition of quantitative hypothesis-testing research in Table 3 .
| Quantitative research hypotheses | |
|---|---|
| Simple hypothesis | |
| - Predicts relationship between single dependent variable and single independent variable | |
| If the dose of the new medication (single independent variable) is high, blood pressure (single dependent variable) is lowered. | |
| Complex hypothesis | |
| - Foretells relationship between two or more independent and dependent variables | |
| The higher the use of anticancer drugs, radiation therapy, and adjunctive agents (3 independent variables), the higher would be the survival rate (1 dependent variable). | |
| Directional hypothesis | |
| - Identifies study direction based on theory towards particular outcome to clarify relationship between variables | |
| Privately funded research projects will have a larger international scope (study direction) than publicly funded research projects. | |
| Non-directional hypothesis | |
| - Nature of relationship between two variables or exact study direction is not identified | |
| - Does not involve a theory | |
| Women and men are different in terms of helpfulness. (Exact study direction is not identified) | |
| Associative hypothesis | |
| - Describes variable interdependency | |
| - Change in one variable causes change in another variable | |
| A larger number of people vaccinated against COVID-19 in the region (change in independent variable) will reduce the region’s incidence of COVID-19 infection (change in dependent variable). | |
| Causal hypothesis | |
| - An effect on dependent variable is predicted from manipulation of independent variable | |
| A change into a high-fiber diet (independent variable) will reduce the blood sugar level (dependent variable) of the patient. | |
| Null hypothesis | |
| - A negative statement indicating no relationship or difference between 2 variables | |
| There is no significant difference in the severity of pulmonary metastases between the new drug (variable 1) and the current drug (variable 2). | |
| Alternative hypothesis | |
| - Following a null hypothesis, an alternative hypothesis predicts a relationship between 2 study variables | |
| The new drug (variable 1) is better on average in reducing the level of pain from pulmonary metastasis than the current drug (variable 2). | |
| Working hypothesis | |
| - A hypothesis that is initially accepted for further research to produce a feasible theory | |
| Dairy cows fed with concentrates of different formulations will produce different amounts of milk. | |
| Statistical hypothesis | |
| - Assumption about the value of population parameter or relationship among several population characteristics | |
| - Validity tested by a statistical experiment or analysis | |
| The mean recovery rate from COVID-19 infection (value of population parameter) is not significantly different between population 1 and population 2. | |
| There is a positive correlation between the level of stress at the workplace and the number of suicides (population characteristics) among working people in Japan. | |
| Logical hypothesis | |
| - Offers or proposes an explanation with limited or no extensive evidence | |
| If healthcare workers provide more educational programs about contraception methods, the number of adolescent pregnancies will be less. | |
| Hypothesis-testing (Quantitative hypothesis-testing research) | |
| - Quantitative research uses deductive reasoning. | |
| - This involves the formation of a hypothesis, collection of data in the investigation of the problem, analysis and use of the data from the investigation, and drawing of conclusions to validate or nullify the hypotheses. | |
Research questions in qualitative research
Unlike research questions in quantitative research, research questions in qualitative research are usually continuously reviewed and reformulated. The central question and associated subquestions are stated more than the hypotheses. 15 The central question broadly explores a complex set of factors surrounding the central phenomenon, aiming to present the varied perspectives of participants. 15
There are varied goals for which qualitative research questions are developed. These questions can function in several ways, such as to 1) identify and describe existing conditions ( contextual research question s); 2) describe a phenomenon ( descriptive research questions ); 3) assess the effectiveness of existing methods, protocols, theories, or procedures ( evaluation research questions ); 4) examine a phenomenon or analyze the reasons or relationships between subjects or phenomena ( explanatory research questions ); or 5) focus on unknown aspects of a particular topic ( exploratory research questions ). 5 In addition, some qualitative research questions provide new ideas for the development of theories and actions ( generative research questions ) or advance specific ideologies of a position ( ideological research questions ). 1 Other qualitative research questions may build on a body of existing literature and become working guidelines ( ethnographic research questions ). Research questions may also be broadly stated without specific reference to the existing literature or a typology of questions ( phenomenological research questions ), may be directed towards generating a theory of some process ( grounded theory questions ), or may address a description of the case and the emerging themes ( qualitative case study questions ). 15 We provide examples of contextual, descriptive, evaluation, explanatory, exploratory, generative, ideological, ethnographic, phenomenological, grounded theory, and qualitative case study research questions in qualitative research in Table 4 , and the definition of qualitative hypothesis-generating research in Table 5 .
| Qualitative research questions | |
|---|---|
| Contextual research question | |
| - Ask the nature of what already exists | |
| - Individuals or groups function to further clarify and understand the natural context of real-world problems | |
| What are the experiences of nurses working night shifts in healthcare during the COVID-19 pandemic? (natural context of real-world problems) | |
| Descriptive research question | |
| - Aims to describe a phenomenon | |
| What are the different forms of disrespect and abuse (phenomenon) experienced by Tanzanian women when giving birth in healthcare facilities? | |
| Evaluation research question | |
| - Examines the effectiveness of existing practice or accepted frameworks | |
| How effective are decision aids (effectiveness of existing practice) in helping decide whether to give birth at home or in a healthcare facility? | |
| Explanatory research question | |
| - Clarifies a previously studied phenomenon and explains why it occurs | |
| Why is there an increase in teenage pregnancy (phenomenon) in Tanzania? | |
| Exploratory research question | |
| - Explores areas that have not been fully investigated to have a deeper understanding of the research problem | |
| What factors affect the mental health of medical students (areas that have not yet been fully investigated) during the COVID-19 pandemic? | |
| Generative research question | |
| - Develops an in-depth understanding of people’s behavior by asking ‘how would’ or ‘what if’ to identify problems and find solutions | |
| How would the extensive research experience of the behavior of new staff impact the success of the novel drug initiative? | |
| Ideological research question | |
| - Aims to advance specific ideas or ideologies of a position | |
| Are Japanese nurses who volunteer in remote African hospitals able to promote humanized care of patients (specific ideas or ideologies) in the areas of safe patient environment, respect of patient privacy, and provision of accurate information related to health and care? | |
| Ethnographic research question | |
| - Clarifies peoples’ nature, activities, their interactions, and the outcomes of their actions in specific settings | |
| What are the demographic characteristics, rehabilitative treatments, community interactions, and disease outcomes (nature, activities, their interactions, and the outcomes) of people in China who are suffering from pneumoconiosis? | |
| Phenomenological research question | |
| - Knows more about the phenomena that have impacted an individual | |
| What are the lived experiences of parents who have been living with and caring for children with a diagnosis of autism? (phenomena that have impacted an individual) | |
| Grounded theory question | |
| - Focuses on social processes asking about what happens and how people interact, or uncovering social relationships and behaviors of groups | |
| What are the problems that pregnant adolescents face in terms of social and cultural norms (social processes), and how can these be addressed? | |
| Qualitative case study question | |
| - Assesses a phenomenon using different sources of data to answer “why” and “how” questions | |
| - Considers how the phenomenon is influenced by its contextual situation. | |
| How does quitting work and assuming the role of a full-time mother (phenomenon assessed) change the lives of women in Japan? | |
| Qualitative research hypotheses | |
|---|---|
| Hypothesis-generating (Qualitative hypothesis-generating research) | |
| - Qualitative research uses inductive reasoning. | |
| - This involves data collection from study participants or the literature regarding a phenomenon of interest, using the collected data to develop a formal hypothesis, and using the formal hypothesis as a framework for testing the hypothesis. | |
| - Qualitative exploratory studies explore areas deeper, clarifying subjective experience and allowing formulation of a formal hypothesis potentially testable in a future quantitative approach. | |
Qualitative studies usually pose at least one central research question and several subquestions starting with How or What . These research questions use exploratory verbs such as explore or describe . These also focus on one central phenomenon of interest, and may mention the participants and research site. 15
Hypotheses in qualitative research
Hypotheses in qualitative research are stated in the form of a clear statement concerning the problem to be investigated. Unlike in quantitative research where hypotheses are usually developed to be tested, qualitative research can lead to both hypothesis-testing and hypothesis-generating outcomes. 2 When studies require both quantitative and qualitative research questions, this suggests an integrative process between both research methods wherein a single mixed-methods research question can be developed. 1
FRAMEWORKS FOR DEVELOPING RESEARCH QUESTIONS AND HYPOTHESES
Research questions followed by hypotheses should be developed before the start of the study. 1 , 12 , 14 It is crucial to develop feasible research questions on a topic that is interesting to both the researcher and the scientific community. This can be achieved by a meticulous review of previous and current studies to establish a novel topic. Specific areas are subsequently focused on to generate ethical research questions. The relevance of the research questions is evaluated in terms of clarity of the resulting data, specificity of the methodology, objectivity of the outcome, depth of the research, and impact of the study. 1 , 5 These aspects constitute the FINER criteria (i.e., Feasible, Interesting, Novel, Ethical, and Relevant). 1 Clarity and effectiveness are achieved if research questions meet the FINER criteria. In addition to the FINER criteria, Ratan et al. described focus, complexity, novelty, feasibility, and measurability for evaluating the effectiveness of research questions. 14
The PICOT and PEO frameworks are also used when developing research questions. 1 The following elements are addressed in these frameworks, PICOT: P-population/patients/problem, I-intervention or indicator being studied, C-comparison group, O-outcome of interest, and T-timeframe of the study; PEO: P-population being studied, E-exposure to preexisting conditions, and O-outcome of interest. 1 Research questions are also considered good if these meet the “FINERMAPS” framework: Feasible, Interesting, Novel, Ethical, Relevant, Manageable, Appropriate, Potential value/publishable, and Systematic. 14
As we indicated earlier, research questions and hypotheses that are not carefully formulated result in unethical studies or poor outcomes. To illustrate this, we provide some examples of ambiguous research question and hypotheses that result in unclear and weak research objectives in quantitative research ( Table 6 ) 16 and qualitative research ( Table 7 ) 17 , and how to transform these ambiguous research question(s) and hypothesis(es) into clear and good statements.
| Variables | Unclear and weak statement (Statement 1) | Clear and good statement (Statement 2) | Points to avoid |
|---|---|---|---|
| Research question | Which is more effective between smoke moxibustion and smokeless moxibustion? | “Moreover, regarding smoke moxibustion versus smokeless moxibustion, it remains unclear which is more effective, safe, and acceptable to pregnant women, and whether there is any difference in the amount of heat generated.” | 1) Vague and unfocused questions |
| 2) Closed questions simply answerable by yes or no | |||
| 3) Questions requiring a simple choice | |||
| Hypothesis | The smoke moxibustion group will have higher cephalic presentation. | “Hypothesis 1. The smoke moxibustion stick group (SM group) and smokeless moxibustion stick group (-SLM group) will have higher rates of cephalic presentation after treatment than the control group. | 1) Unverifiable hypotheses |
| Hypothesis 2. The SM group and SLM group will have higher rates of cephalic presentation at birth than the control group. | 2) Incompletely stated groups of comparison | ||
| Hypothesis 3. There will be no significant differences in the well-being of the mother and child among the three groups in terms of the following outcomes: premature birth, premature rupture of membranes (PROM) at < 37 weeks, Apgar score < 7 at 5 min, umbilical cord blood pH < 7.1, admission to neonatal intensive care unit (NICU), and intrauterine fetal death.” | 3) Insufficiently described variables or outcomes | ||
| Research objective | To determine which is more effective between smoke moxibustion and smokeless moxibustion. | “The specific aims of this pilot study were (a) to compare the effects of smoke moxibustion and smokeless moxibustion treatments with the control group as a possible supplement to ECV for converting breech presentation to cephalic presentation and increasing adherence to the newly obtained cephalic position, and (b) to assess the effects of these treatments on the well-being of the mother and child.” | 1) Poor understanding of the research question and hypotheses |
| 2) Insufficient description of population, variables, or study outcomes |
a These statements were composed for comparison and illustrative purposes only.
b These statements are direct quotes from Higashihara and Horiuchi. 16
| Variables | Unclear and weak statement (Statement 1) | Clear and good statement (Statement 2) | Points to avoid |
|---|---|---|---|
| Research question | Does disrespect and abuse (D&A) occur in childbirth in Tanzania? | How does disrespect and abuse (D&A) occur and what are the types of physical and psychological abuses observed in midwives’ actual care during facility-based childbirth in urban Tanzania? | 1) Ambiguous or oversimplistic questions |
| 2) Questions unverifiable by data collection and analysis | |||
| Hypothesis | Disrespect and abuse (D&A) occur in childbirth in Tanzania. | Hypothesis 1: Several types of physical and psychological abuse by midwives in actual care occur during facility-based childbirth in urban Tanzania. | 1) Statements simply expressing facts |
| Hypothesis 2: Weak nursing and midwifery management contribute to the D&A of women during facility-based childbirth in urban Tanzania. | 2) Insufficiently described concepts or variables | ||
| Research objective | To describe disrespect and abuse (D&A) in childbirth in Tanzania. | “This study aimed to describe from actual observations the respectful and disrespectful care received by women from midwives during their labor period in two hospitals in urban Tanzania.” | 1) Statements unrelated to the research question and hypotheses |
| 2) Unattainable or unexplorable objectives |
a This statement is a direct quote from Shimoda et al. 17
The other statements were composed for comparison and illustrative purposes only.
CONSTRUCTING RESEARCH QUESTIONS AND HYPOTHESES
To construct effective research questions and hypotheses, it is very important to 1) clarify the background and 2) identify the research problem at the outset of the research, within a specific timeframe. 9 Then, 3) review or conduct preliminary research to collect all available knowledge about the possible research questions by studying theories and previous studies. 18 Afterwards, 4) construct research questions to investigate the research problem. Identify variables to be accessed from the research questions 4 and make operational definitions of constructs from the research problem and questions. Thereafter, 5) construct specific deductive or inductive predictions in the form of hypotheses. 4 Finally, 6) state the study aims . This general flow for constructing effective research questions and hypotheses prior to conducting research is shown in Fig. 1 .

Research questions are used more frequently in qualitative research than objectives or hypotheses. 3 These questions seek to discover, understand, explore or describe experiences by asking “What” or “How.” The questions are open-ended to elicit a description rather than to relate variables or compare groups. The questions are continually reviewed, reformulated, and changed during the qualitative study. 3 Research questions are also used more frequently in survey projects than hypotheses in experiments in quantitative research to compare variables and their relationships.
Hypotheses are constructed based on the variables identified and as an if-then statement, following the template, ‘If a specific action is taken, then a certain outcome is expected.’ At this stage, some ideas regarding expectations from the research to be conducted must be drawn. 18 Then, the variables to be manipulated (independent) and influenced (dependent) are defined. 4 Thereafter, the hypothesis is stated and refined, and reproducible data tailored to the hypothesis are identified, collected, and analyzed. 4 The hypotheses must be testable and specific, 18 and should describe the variables and their relationships, the specific group being studied, and the predicted research outcome. 18 Hypotheses construction involves a testable proposition to be deduced from theory, and independent and dependent variables to be separated and measured separately. 3 Therefore, good hypotheses must be based on good research questions constructed at the start of a study or trial. 12
In summary, research questions are constructed after establishing the background of the study. Hypotheses are then developed based on the research questions. Thus, it is crucial to have excellent research questions to generate superior hypotheses. In turn, these would determine the research objectives and the design of the study, and ultimately, the outcome of the research. 12 Algorithms for building research questions and hypotheses are shown in Fig. 2 for quantitative research and in Fig. 3 for qualitative research.

EXAMPLES OF RESEARCH QUESTIONS FROM PUBLISHED ARTICLES
- EXAMPLE 1. Descriptive research question (quantitative research)
- - Presents research variables to be assessed (distinct phenotypes and subphenotypes)
- “BACKGROUND: Since COVID-19 was identified, its clinical and biological heterogeneity has been recognized. Identifying COVID-19 phenotypes might help guide basic, clinical, and translational research efforts.
- RESEARCH QUESTION: Does the clinical spectrum of patients with COVID-19 contain distinct phenotypes and subphenotypes? ” 19
- EXAMPLE 2. Relationship research question (quantitative research)
- - Shows interactions between dependent variable (static postural control) and independent variable (peripheral visual field loss)
- “Background: Integration of visual, vestibular, and proprioceptive sensations contributes to postural control. People with peripheral visual field loss have serious postural instability. However, the directional specificity of postural stability and sensory reweighting caused by gradual peripheral visual field loss remain unclear.
- Research question: What are the effects of peripheral visual field loss on static postural control ?” 20
- EXAMPLE 3. Comparative research question (quantitative research)
- - Clarifies the difference among groups with an outcome variable (patients enrolled in COMPERA with moderate PH or severe PH in COPD) and another group without the outcome variable (patients with idiopathic pulmonary arterial hypertension (IPAH))
- “BACKGROUND: Pulmonary hypertension (PH) in COPD is a poorly investigated clinical condition.
- RESEARCH QUESTION: Which factors determine the outcome of PH in COPD?
- STUDY DESIGN AND METHODS: We analyzed the characteristics and outcome of patients enrolled in the Comparative, Prospective Registry of Newly Initiated Therapies for Pulmonary Hypertension (COMPERA) with moderate or severe PH in COPD as defined during the 6th PH World Symposium who received medical therapy for PH and compared them with patients with idiopathic pulmonary arterial hypertension (IPAH) .” 21
- EXAMPLE 4. Exploratory research question (qualitative research)
- - Explores areas that have not been fully investigated (perspectives of families and children who receive care in clinic-based child obesity treatment) to have a deeper understanding of the research problem
- “Problem: Interventions for children with obesity lead to only modest improvements in BMI and long-term outcomes, and data are limited on the perspectives of families of children with obesity in clinic-based treatment. This scoping review seeks to answer the question: What is known about the perspectives of families and children who receive care in clinic-based child obesity treatment? This review aims to explore the scope of perspectives reported by families of children with obesity who have received individualized outpatient clinic-based obesity treatment.” 22
- EXAMPLE 5. Relationship research question (quantitative research)
- - Defines interactions between dependent variable (use of ankle strategies) and independent variable (changes in muscle tone)
- “Background: To maintain an upright standing posture against external disturbances, the human body mainly employs two types of postural control strategies: “ankle strategy” and “hip strategy.” While it has been reported that the magnitude of the disturbance alters the use of postural control strategies, it has not been elucidated how the level of muscle tone, one of the crucial parameters of bodily function, determines the use of each strategy. We have previously confirmed using forward dynamics simulations of human musculoskeletal models that an increased muscle tone promotes the use of ankle strategies. The objective of the present study was to experimentally evaluate a hypothesis: an increased muscle tone promotes the use of ankle strategies. Research question: Do changes in the muscle tone affect the use of ankle strategies ?” 23
EXAMPLES OF HYPOTHESES IN PUBLISHED ARTICLES
- EXAMPLE 1. Working hypothesis (quantitative research)
- - A hypothesis that is initially accepted for further research to produce a feasible theory
- “As fever may have benefit in shortening the duration of viral illness, it is plausible to hypothesize that the antipyretic efficacy of ibuprofen may be hindering the benefits of a fever response when taken during the early stages of COVID-19 illness .” 24
- “In conclusion, it is plausible to hypothesize that the antipyretic efficacy of ibuprofen may be hindering the benefits of a fever response . The difference in perceived safety of these agents in COVID-19 illness could be related to the more potent efficacy to reduce fever with ibuprofen compared to acetaminophen. Compelling data on the benefit of fever warrant further research and review to determine when to treat or withhold ibuprofen for early stage fever for COVID-19 and other related viral illnesses .” 24
- EXAMPLE 2. Exploratory hypothesis (qualitative research)
- - Explores particular areas deeper to clarify subjective experience and develop a formal hypothesis potentially testable in a future quantitative approach
- “We hypothesized that when thinking about a past experience of help-seeking, a self distancing prompt would cause increased help-seeking intentions and more favorable help-seeking outcome expectations .” 25
- “Conclusion
- Although a priori hypotheses were not supported, further research is warranted as results indicate the potential for using self-distancing approaches to increasing help-seeking among some people with depressive symptomatology.” 25
- EXAMPLE 3. Hypothesis-generating research to establish a framework for hypothesis testing (qualitative research)
- “We hypothesize that compassionate care is beneficial for patients (better outcomes), healthcare systems and payers (lower costs), and healthcare providers (lower burnout). ” 26
- Compassionomics is the branch of knowledge and scientific study of the effects of compassionate healthcare. Our main hypotheses are that compassionate healthcare is beneficial for (1) patients, by improving clinical outcomes, (2) healthcare systems and payers, by supporting financial sustainability, and (3) HCPs, by lowering burnout and promoting resilience and well-being. The purpose of this paper is to establish a scientific framework for testing the hypotheses above . If these hypotheses are confirmed through rigorous research, compassionomics will belong in the science of evidence-based medicine, with major implications for all healthcare domains.” 26
- EXAMPLE 4. Statistical hypothesis (quantitative research)
- - An assumption is made about the relationship among several population characteristics ( gender differences in sociodemographic and clinical characteristics of adults with ADHD ). Validity is tested by statistical experiment or analysis ( chi-square test, Students t-test, and logistic regression analysis)
- “Our research investigated gender differences in sociodemographic and clinical characteristics of adults with ADHD in a Japanese clinical sample. Due to unique Japanese cultural ideals and expectations of women's behavior that are in opposition to ADHD symptoms, we hypothesized that women with ADHD experience more difficulties and present more dysfunctions than men . We tested the following hypotheses: first, women with ADHD have more comorbidities than men with ADHD; second, women with ADHD experience more social hardships than men, such as having less full-time employment and being more likely to be divorced.” 27
- “Statistical Analysis
- ( text omitted ) Between-gender comparisons were made using the chi-squared test for categorical variables and Students t-test for continuous variables…( text omitted ). A logistic regression analysis was performed for employment status, marital status, and comorbidity to evaluate the independent effects of gender on these dependent variables.” 27
EXAMPLES OF HYPOTHESIS AS WRITTEN IN PUBLISHED ARTICLES IN RELATION TO OTHER PARTS
- EXAMPLE 1. Background, hypotheses, and aims are provided
- “Pregnant women need skilled care during pregnancy and childbirth, but that skilled care is often delayed in some countries …( text omitted ). The focused antenatal care (FANC) model of WHO recommends that nurses provide information or counseling to all pregnant women …( text omitted ). Job aids are visual support materials that provide the right kind of information using graphics and words in a simple and yet effective manner. When nurses are not highly trained or have many work details to attend to, these job aids can serve as a content reminder for the nurses and can be used for educating their patients (Jennings, Yebadokpo, Affo, & Agbogbe, 2010) ( text omitted ). Importantly, additional evidence is needed to confirm how job aids can further improve the quality of ANC counseling by health workers in maternal care …( text omitted )” 28
- “ This has led us to hypothesize that the quality of ANC counseling would be better if supported by job aids. Consequently, a better quality of ANC counseling is expected to produce higher levels of awareness concerning the danger signs of pregnancy and a more favorable impression of the caring behavior of nurses .” 28
- “This study aimed to examine the differences in the responses of pregnant women to a job aid-supported intervention during ANC visit in terms of 1) their understanding of the danger signs of pregnancy and 2) their impression of the caring behaviors of nurses to pregnant women in rural Tanzania.” 28
- EXAMPLE 2. Background, hypotheses, and aims are provided
- “We conducted a two-arm randomized controlled trial (RCT) to evaluate and compare changes in salivary cortisol and oxytocin levels of first-time pregnant women between experimental and control groups. The women in the experimental group touched and held an infant for 30 min (experimental intervention protocol), whereas those in the control group watched a DVD movie of an infant (control intervention protocol). The primary outcome was salivary cortisol level and the secondary outcome was salivary oxytocin level.” 29
- “ We hypothesize that at 30 min after touching and holding an infant, the salivary cortisol level will significantly decrease and the salivary oxytocin level will increase in the experimental group compared with the control group .” 29
- EXAMPLE 3. Background, aim, and hypothesis are provided
- “In countries where the maternal mortality ratio remains high, antenatal education to increase Birth Preparedness and Complication Readiness (BPCR) is considered one of the top priorities [1]. BPCR includes birth plans during the antenatal period, such as the birthplace, birth attendant, transportation, health facility for complications, expenses, and birth materials, as well as family coordination to achieve such birth plans. In Tanzania, although increasing, only about half of all pregnant women attend an antenatal clinic more than four times [4]. Moreover, the information provided during antenatal care (ANC) is insufficient. In the resource-poor settings, antenatal group education is a potential approach because of the limited time for individual counseling at antenatal clinics.” 30
- “This study aimed to evaluate an antenatal group education program among pregnant women and their families with respect to birth-preparedness and maternal and infant outcomes in rural villages of Tanzania.” 30
- “ The study hypothesis was if Tanzanian pregnant women and their families received a family-oriented antenatal group education, they would (1) have a higher level of BPCR, (2) attend antenatal clinic four or more times, (3) give birth in a health facility, (4) have less complications of women at birth, and (5) have less complications and deaths of infants than those who did not receive the education .” 30
Research questions and hypotheses are crucial components to any type of research, whether quantitative or qualitative. These questions should be developed at the very beginning of the study. Excellent research questions lead to superior hypotheses, which, like a compass, set the direction of research, and can often determine the successful conduct of the study. Many research studies have floundered because the development of research questions and subsequent hypotheses was not given the thought and meticulous attention needed. The development of research questions and hypotheses is an iterative process based on extensive knowledge of the literature and insightful grasp of the knowledge gap. Focused, concise, and specific research questions provide a strong foundation for constructing hypotheses which serve as formal predictions about the research outcomes. Research questions and hypotheses are crucial elements of research that should not be overlooked. They should be carefully thought of and constructed when planning research. This avoids unethical studies and poor outcomes by defining well-founded objectives that determine the design, course, and outcome of the study.
Disclosure: The authors have no potential conflicts of interest to disclose.
Author Contributions:
- Conceptualization: Barroga E, Matanguihan GJ.
- Methodology: Barroga E, Matanguihan GJ.
- Writing - original draft: Barroga E, Matanguihan GJ.
- Writing - review & editing: Barroga E, Matanguihan GJ.
Ohio State nav bar
The Ohio State University
- BuckeyeLink
- Find People
- Search Ohio State
Research Questions & Hypotheses
Generally, in quantitative studies, reviewers expect hypotheses rather than research questions. However, both research questions and hypotheses serve different purposes and can be beneficial when used together.
Research Questions
Clarify the research’s aim (farrugia et al., 2010).
- Research often begins with an interest in a topic, but a deep understanding of the subject is crucial to formulate an appropriate research question.
- Descriptive: “What factors most influence the academic achievement of senior high school students?”
- Comparative: “What is the performance difference between teaching methods A and B?”
- Relationship-based: “What is the relationship between self-efficacy and academic achievement?”
- Increasing knowledge about a subject can be achieved through systematic literature reviews, in-depth interviews with patients (and proxies), focus groups, and consultations with field experts.
- Some funding bodies, like the Canadian Institute for Health Research, recommend conducting a systematic review or a pilot study before seeking grants for full trials.
- The presence of multiple research questions in a study can complicate the design, statistical analysis, and feasibility.
- It’s advisable to focus on a single primary research question for the study.
- The primary question, clearly stated at the end of a grant proposal’s introduction, usually specifies the study population, intervention, and other relevant factors.
- The FINER criteria underscore aspects that can enhance the chances of a successful research project, including specifying the population of interest, aligning with scientific and public interest, clinical relevance, and contribution to the field, while complying with ethical and national research standards.
| Feasible | ||
| Interesting | ||
| Novel | ||
| Ethical | ||
| Relevant |
- The P ICOT approach is crucial in developing the study’s framework and protocol, influencing inclusion and exclusion criteria and identifying patient groups for inclusion.
| Population (patients) | ||
| Intervention (for intervention studies only) | ||
| Comparison group | ||
| Outcome of interest | ||
| Time |
- Defining the specific population, intervention, comparator, and outcome helps in selecting the right outcome measurement tool.
- The more precise the population definition and stricter the inclusion and exclusion criteria, the more significant the impact on the interpretation, applicability, and generalizability of the research findings.
- A restricted study population enhances internal validity but may limit the study’s external validity and generalizability to clinical practice.
- A broadly defined study population may better reflect clinical practice but could increase bias and reduce internal validity.
- An inadequately formulated research question can negatively impact study design, potentially leading to ineffective outcomes and affecting publication prospects.
Checklist: Good research questions for social science projects (Panke, 2018)
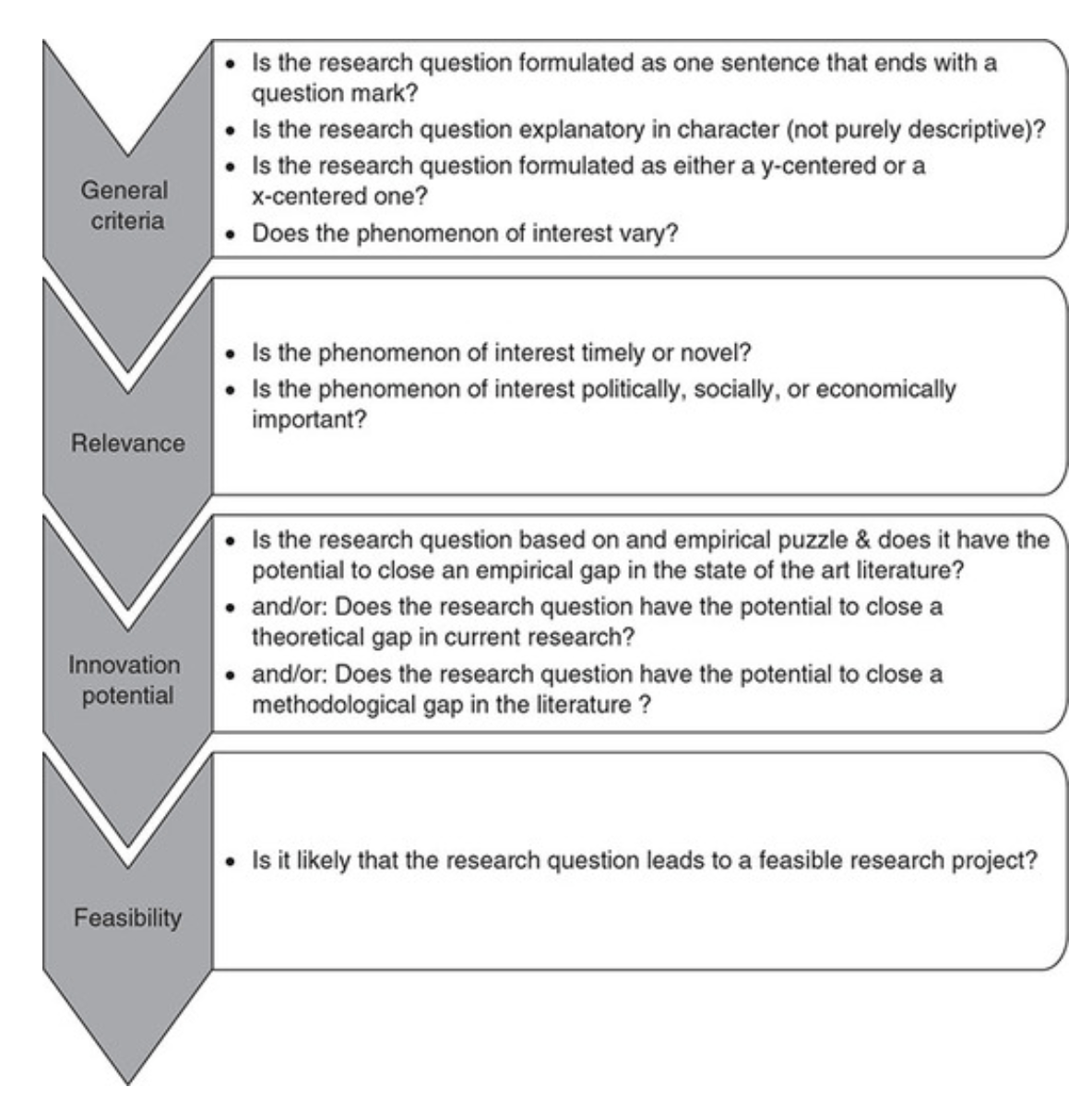
Research Hypotheses
Present the researcher’s predictions based on specific statements.
- These statements define the research problem or issue and indicate the direction of the researcher’s predictions.
- Formulating the research question and hypothesis from existing data (e.g., a database) can lead to multiple statistical comparisons and potentially spurious findings due to chance.
- The research or clinical hypothesis, derived from the research question, shapes the study’s key elements: sampling strategy, intervention, comparison, and outcome variables.
- Hypotheses can express a single outcome or multiple outcomes.
- After statistical testing, the null hypothesis is either rejected or not rejected based on whether the study’s findings are statistically significant.
- Hypothesis testing helps determine if observed findings are due to true differences and not chance.
- Hypotheses can be 1-sided (specific direction of difference) or 2-sided (presence of a difference without specifying direction).
- 2-sided hypotheses are generally preferred unless there’s a strong justification for a 1-sided hypothesis.
- A solid research hypothesis, informed by a good research question, influences the research design and paves the way for defining clear research objectives.
Types of Research Hypothesis
- In a Y-centered research design, the focus is on the dependent variable (DV) which is specified in the research question. Theories are then used to identify independent variables (IV) and explain their causal relationship with the DV.
- Example: “An increase in teacher-led instructional time (IV) is likely to improve student reading comprehension scores (DV), because extensive guided practice under expert supervision enhances learning retention and skill mastery.”
- Hypothesis Explanation: The dependent variable (student reading comprehension scores) is the focus, and the hypothesis explores how changes in the independent variable (teacher-led instructional time) affect it.
- In X-centered research designs, the independent variable is specified in the research question. Theories are used to determine potential dependent variables and the causal mechanisms at play.
- Example: “Implementing technology-based learning tools (IV) is likely to enhance student engagement in the classroom (DV), because interactive and multimedia content increases student interest and participation.”
- Hypothesis Explanation: The independent variable (technology-based learning tools) is the focus, with the hypothesis exploring its impact on a potential dependent variable (student engagement).
- Probabilistic hypotheses suggest that changes in the independent variable are likely to lead to changes in the dependent variable in a predictable manner, but not with absolute certainty.
- Example: “The more teachers engage in professional development programs (IV), the more their teaching effectiveness (DV) is likely to improve, because continuous training updates pedagogical skills and knowledge.”
- Hypothesis Explanation: This hypothesis implies a probable relationship between the extent of professional development (IV) and teaching effectiveness (DV).
- Deterministic hypotheses state that a specific change in the independent variable will lead to a specific change in the dependent variable, implying a more direct and certain relationship.
- Example: “If the school curriculum changes from traditional lecture-based methods to project-based learning (IV), then student collaboration skills (DV) are expected to improve because project-based learning inherently requires teamwork and peer interaction.”
- Hypothesis Explanation: This hypothesis presumes a direct and definite outcome (improvement in collaboration skills) resulting from a specific change in the teaching method.
- Example : “Students who identify as visual learners will score higher on tests that are presented in a visually rich format compared to tests presented in a text-only format.”
- Explanation : This hypothesis aims to describe the potential difference in test scores between visual learners taking visually rich tests and text-only tests, without implying a direct cause-and-effect relationship.
- Example : “Teaching method A will improve student performance more than method B.”
- Explanation : This hypothesis compares the effectiveness of two different teaching methods, suggesting that one will lead to better student performance than the other. It implies a direct comparison but does not necessarily establish a causal mechanism.
- Example : “Students with higher self-efficacy will show higher levels of academic achievement.”
- Explanation : This hypothesis predicts a relationship between the variable of self-efficacy and academic achievement. Unlike a causal hypothesis, it does not necessarily suggest that one variable causes changes in the other, but rather that they are related in some way.
Tips for developing research questions and hypotheses for research studies
- Perform a systematic literature review (if one has not been done) to increase knowledge and familiarity with the topic and to assist with research development.
- Learn about current trends and technological advances on the topic.
- Seek careful input from experts, mentors, colleagues, and collaborators to refine your research question as this will aid in developing the research question and guide the research study.
- Use the FINER criteria in the development of the research question.
- Ensure that the research question follows PICOT format.
- Develop a research hypothesis from the research question.
- Ensure that the research question and objectives are answerable, feasible, and clinically relevant.
If your research hypotheses are derived from your research questions, particularly when multiple hypotheses address a single question, it’s recommended to use both research questions and hypotheses. However, if this isn’t the case, using hypotheses over research questions is advised. It’s important to note these are general guidelines, not strict rules. If you opt not to use hypotheses, consult with your supervisor for the best approach.
Farrugia, P., Petrisor, B. A., Farrokhyar, F., & Bhandari, M. (2010). Practical tips for surgical research: Research questions, hypotheses and objectives. Canadian journal of surgery. Journal canadien de chirurgie , 53 (4), 278–281.
Hulley, S. B., Cummings, S. R., Browner, W. S., Grady, D., & Newman, T. B. (2007). Designing clinical research. Philadelphia.
Panke, D. (2018). Research design & method selection: Making good choices in the social sciences. Research Design & Method Selection , 1-368.
- Privacy Policy

Home » Research Objectives – Types, Examples and Writing Guide
Research Objectives – Types, Examples and Writing Guide
Table of Contents

Research Objectives
Research objectives refer to the specific goals or aims of a research study. They provide a clear and concise description of what the researcher hopes to achieve by conducting the research . The objectives are typically based on the research questions and hypotheses formulated at the beginning of the study and are used to guide the research process.
Types of Research Objectives
Here are the different types of research objectives in research:
- Exploratory Objectives: These objectives are used to explore a topic, issue, or phenomenon that has not been studied in-depth before. The aim of exploratory research is to gain a better understanding of the subject matter and generate new ideas and hypotheses .
- Descriptive Objectives: These objectives aim to describe the characteristics, features, or attributes of a particular population, group, or phenomenon. Descriptive research answers the “what” questions and provides a snapshot of the subject matter.
- Explanatory Objectives : These objectives aim to explain the relationships between variables or factors. Explanatory research seeks to identify the cause-and-effect relationships between different phenomena.
- Predictive Objectives: These objectives aim to predict future events or outcomes based on existing data or trends. Predictive research uses statistical models to forecast future trends or outcomes.
- Evaluative Objectives : These objectives aim to evaluate the effectiveness or impact of a program, intervention, or policy. Evaluative research seeks to assess the outcomes or results of a particular intervention or program.
- Prescriptive Objectives: These objectives aim to provide recommendations or solutions to a particular problem or issue. Prescriptive research identifies the best course of action based on the results of the study.
- Diagnostic Objectives : These objectives aim to identify the causes or factors contributing to a particular problem or issue. Diagnostic research seeks to uncover the underlying reasons for a particular phenomenon.
- Comparative Objectives: These objectives aim to compare two or more groups, populations, or phenomena to identify similarities and differences. Comparative research is used to determine which group or approach is more effective or has better outcomes.
- Historical Objectives: These objectives aim to examine past events, trends, or phenomena to gain a better understanding of their significance and impact. Historical research uses archival data, documents, and records to study past events.
- Ethnographic Objectives : These objectives aim to understand the culture, beliefs, and practices of a particular group or community. Ethnographic research involves immersive fieldwork and observation to gain an insider’s perspective of the group being studied.
- Action-oriented Objectives: These objectives aim to bring about social or organizational change. Action-oriented research seeks to identify practical solutions to social problems and to promote positive change in society.
- Conceptual Objectives: These objectives aim to develop new theories, models, or frameworks to explain a particular phenomenon or set of phenomena. Conceptual research seeks to provide a deeper understanding of the subject matter by developing new theoretical perspectives.
- Methodological Objectives: These objectives aim to develop and improve research methods and techniques. Methodological research seeks to advance the field of research by improving the validity, reliability, and accuracy of research methods and tools.
- Theoretical Objectives : These objectives aim to test and refine existing theories or to develop new theoretical perspectives. Theoretical research seeks to advance the field of knowledge by testing and refining existing theories or by developing new theoretical frameworks.
- Measurement Objectives : These objectives aim to develop and validate measurement instruments, such as surveys, questionnaires, and tests. Measurement research seeks to improve the quality and reliability of data collection and analysis by developing and testing new measurement tools.
- Design Objectives : These objectives aim to develop and refine research designs, such as experimental, quasi-experimental, and observational designs. Design research seeks to improve the quality and validity of research by developing and testing new research designs.
- Sampling Objectives: These objectives aim to develop and refine sampling techniques, such as probability and non-probability sampling methods. Sampling research seeks to improve the representativeness and generalizability of research findings by developing and testing new sampling techniques.
How to Write Research Objectives
Writing clear and concise research objectives is an important part of any research project, as it helps to guide the study and ensure that it is focused and relevant. Here are some steps to follow when writing research objectives:
- Identify the research problem : Before you can write research objectives, you need to identify the research problem you are trying to address. This should be a clear and specific problem that can be addressed through research.
- Define the research questions : Based on the research problem, define the research questions you want to answer. These questions should be specific and should guide the research process.
- Identify the variables : Identify the key variables that you will be studying in your research. These are the factors that you will be measuring, manipulating, or analyzing to answer your research questions.
- Write specific objectives: Write specific, measurable objectives that will help you answer your research questions. These objectives should be clear and concise and should indicate what you hope to achieve through your research.
- Use the SMART criteria: To ensure that your research objectives are well-defined and achievable, use the SMART criteria. This means that your objectives should be Specific, Measurable, Achievable, Relevant, and Time-bound.
- Revise and refine: Once you have written your research objectives, revise and refine them to ensure that they are clear, concise, and achievable. Make sure that they align with your research questions and variables, and that they will help you answer your research problem.
Example of Research Objectives
Examples of research objectives Could be:
Research Objectives for the topic of “The Impact of Artificial Intelligence on Employment”:
- To investigate the effects of the adoption of AI on employment trends across various industries and occupations.
- To explore the potential for AI to create new job opportunities and transform existing roles in the workforce.
- To examine the social and economic implications of the widespread use of AI for employment, including issues such as income inequality and access to education and training.
- To identify the skills and competencies that will be required for individuals to thrive in an AI-driven workplace, and to explore the role of education and training in developing these skills.
- To evaluate the ethical and legal considerations surrounding the use of AI for employment, including issues such as bias, privacy, and the responsibility of employers and policymakers to protect workers’ rights.
When to Write Research Objectives
- At the beginning of a research project : Research objectives should be identified and written down before starting a research project. This helps to ensure that the project is focused and that data collection and analysis efforts are aligned with the intended purpose of the research.
- When refining research questions: Writing research objectives can help to clarify and refine research questions. Objectives provide a more concrete and specific framework for addressing research questions, which can improve the overall quality and direction of a research project.
- After conducting a literature review : Conducting a literature review can help to identify gaps in knowledge and areas that require further research. Writing research objectives can help to define and focus the research effort in these areas.
- When developing a research proposal: Research objectives are an important component of a research proposal. They help to articulate the purpose and scope of the research, and provide a clear and concise summary of the expected outcomes and contributions of the research.
- When seeking funding for research: Funding agencies often require a detailed description of research objectives as part of a funding proposal. Writing clear and specific research objectives can help to demonstrate the significance and potential impact of a research project, and increase the chances of securing funding.
- When designing a research study : Research objectives guide the design and implementation of a research study. They help to identify the appropriate research methods, sampling strategies, data collection and analysis techniques, and other relevant aspects of the study design.
- When communicating research findings: Research objectives provide a clear and concise summary of the main research questions and outcomes. They are often included in research reports and publications, and can help to ensure that the research findings are communicated effectively and accurately to a wide range of audiences.
- When evaluating research outcomes : Research objectives provide a basis for evaluating the success of a research project. They help to measure the degree to which research questions have been answered and the extent to which research outcomes have been achieved.
- When conducting research in a team : Writing research objectives can facilitate communication and collaboration within a research team. Objectives provide a shared understanding of the research purpose and goals, and can help to ensure that team members are working towards a common objective.
Purpose of Research Objectives
Some of the main purposes of research objectives include:
- To clarify the research question or problem : Research objectives help to define the specific aspects of the research question or problem that the study aims to address. This makes it easier to design a study that is focused and relevant.
- To guide the research design: Research objectives help to determine the research design, including the research methods, data collection techniques, and sampling strategy. This ensures that the study is structured and efficient.
- To measure progress : Research objectives provide a way to measure progress throughout the research process. They help the researcher to evaluate whether they are on track and meeting their goals.
- To communicate the research goals : Research objectives provide a clear and concise description of the research goals. This helps to communicate the purpose of the study to other researchers, stakeholders, and the general public.
Advantages of Research Objectives
Here are some advantages of having well-defined research objectives:
- Focus : Research objectives help to focus the research effort on specific areas of inquiry. By identifying clear research questions, the researcher can narrow down the scope of the study and avoid getting sidetracked by irrelevant information.
- Clarity : Clearly stated research objectives provide a roadmap for the research study. They provide a clear direction for the research, making it easier for the researcher to stay on track and achieve their goals.
- Measurability : Well-defined research objectives provide measurable outcomes that can be used to evaluate the success of the research project. This helps to ensure that the research is effective and that the research goals are achieved.
- Feasibility : Research objectives help to ensure that the research project is feasible. By clearly defining the research goals, the researcher can identify the resources required to achieve those goals and determine whether those resources are available.
- Relevance : Research objectives help to ensure that the research study is relevant and meaningful. By identifying specific research questions, the researcher can ensure that the study addresses important issues and contributes to the existing body of knowledge.
About the author
Muhammad Hassan
Researcher, Academic Writer, Web developer
You may also like

Research Paper Conclusion – Writing Guide and...

Data Analysis – Process, Methods and Types

Theoretical Framework – Types, Examples and...

Conceptual Framework – Types, Methodology and...

Research Paper Abstract – Writing Guide and...

Table of Contents – Types, Formats, Examples
Educational resources and simple solutions for your research journey

What is a Research Hypothesis: How to Write it, Types, and Examples

Any research begins with a research question and a research hypothesis . A research question alone may not suffice to design the experiment(s) needed to answer it. A hypothesis is central to the scientific method. But what is a hypothesis ? A hypothesis is a testable statement that proposes a possible explanation to a phenomenon, and it may include a prediction. Next, you may ask what is a research hypothesis ? Simply put, a research hypothesis is a prediction or educated guess about the relationship between the variables that you want to investigate.
It is important to be thorough when developing your research hypothesis. Shortcomings in the framing of a hypothesis can affect the study design and the results. A better understanding of the research hypothesis definition and characteristics of a good hypothesis will make it easier for you to develop your own hypothesis for your research. Let’s dive in to know more about the types of research hypothesis , how to write a research hypothesis , and some research hypothesis examples .
Table of Contents
What is a hypothesis ?
A hypothesis is based on the existing body of knowledge in a study area. Framed before the data are collected, a hypothesis states the tentative relationship between independent and dependent variables, along with a prediction of the outcome.
What is a research hypothesis ?
Young researchers starting out their journey are usually brimming with questions like “ What is a hypothesis ?” “ What is a research hypothesis ?” “How can I write a good research hypothesis ?”
A research hypothesis is a statement that proposes a possible explanation for an observable phenomenon or pattern. It guides the direction of a study and predicts the outcome of the investigation. A research hypothesis is testable, i.e., it can be supported or disproven through experimentation or observation.

Characteristics of a good hypothesis
Here are the characteristics of a good hypothesis :
- Clearly formulated and free of language errors and ambiguity
- Concise and not unnecessarily verbose
- Has clearly defined variables
- Testable and stated in a way that allows for it to be disproven
- Can be tested using a research design that is feasible, ethical, and practical
- Specific and relevant to the research problem
- Rooted in a thorough literature search
- Can generate new knowledge or understanding.
How to create an effective research hypothesis
A study begins with the formulation of a research question. A researcher then performs background research. This background information forms the basis for building a good research hypothesis . The researcher then performs experiments, collects, and analyzes the data, interprets the findings, and ultimately, determines if the findings support or negate the original hypothesis.
Let’s look at each step for creating an effective, testable, and good research hypothesis :
- Identify a research problem or question: Start by identifying a specific research problem.
- Review the literature: Conduct an in-depth review of the existing literature related to the research problem to grasp the current knowledge and gaps in the field.
- Formulate a clear and testable hypothesis : Based on the research question, use existing knowledge to form a clear and testable hypothesis . The hypothesis should state a predicted relationship between two or more variables that can be measured and manipulated. Improve the original draft till it is clear and meaningful.
- State the null hypothesis: The null hypothesis is a statement that there is no relationship between the variables you are studying.
- Define the population and sample: Clearly define the population you are studying and the sample you will be using for your research.
- Select appropriate methods for testing the hypothesis: Select appropriate research methods, such as experiments, surveys, or observational studies, which will allow you to test your research hypothesis .
Remember that creating a research hypothesis is an iterative process, i.e., you might have to revise it based on the data you collect. You may need to test and reject several hypotheses before answering the research problem.
How to write a research hypothesis
When you start writing a research hypothesis , you use an “if–then” statement format, which states the predicted relationship between two or more variables. Clearly identify the independent variables (the variables being changed) and the dependent variables (the variables being measured), as well as the population you are studying. Review and revise your hypothesis as needed.
An example of a research hypothesis in this format is as follows:
“ If [athletes] follow [cold water showers daily], then their [endurance] increases.”
Population: athletes
Independent variable: daily cold water showers
Dependent variable: endurance
You may have understood the characteristics of a good hypothesis . But note that a research hypothesis is not always confirmed; a researcher should be prepared to accept or reject the hypothesis based on the study findings.

Research hypothesis checklist
Following from above, here is a 10-point checklist for a good research hypothesis :
- Testable: A research hypothesis should be able to be tested via experimentation or observation.
- Specific: A research hypothesis should clearly state the relationship between the variables being studied.
- Based on prior research: A research hypothesis should be based on existing knowledge and previous research in the field.
- Falsifiable: A research hypothesis should be able to be disproven through testing.
- Clear and concise: A research hypothesis should be stated in a clear and concise manner.
- Logical: A research hypothesis should be logical and consistent with current understanding of the subject.
- Relevant: A research hypothesis should be relevant to the research question and objectives.
- Feasible: A research hypothesis should be feasible to test within the scope of the study.
- Reflects the population: A research hypothesis should consider the population or sample being studied.
- Uncomplicated: A good research hypothesis is written in a way that is easy for the target audience to understand.
By following this research hypothesis checklist , you will be able to create a research hypothesis that is strong, well-constructed, and more likely to yield meaningful results.

Types of research hypothesis
Different types of research hypothesis are used in scientific research:
1. Null hypothesis:
A null hypothesis states that there is no change in the dependent variable due to changes to the independent variable. This means that the results are due to chance and are not significant. A null hypothesis is denoted as H0 and is stated as the opposite of what the alternative hypothesis states.
Example: “ The newly identified virus is not zoonotic .”
2. Alternative hypothesis:
This states that there is a significant difference or relationship between the variables being studied. It is denoted as H1 or Ha and is usually accepted or rejected in favor of the null hypothesis.
Example: “ The newly identified virus is zoonotic .”
3. Directional hypothesis :
This specifies the direction of the relationship or difference between variables; therefore, it tends to use terms like increase, decrease, positive, negative, more, or less.
Example: “ The inclusion of intervention X decreases infant mortality compared to the original treatment .”
4. Non-directional hypothesis:
While it does not predict the exact direction or nature of the relationship between the two variables, a non-directional hypothesis states the existence of a relationship or difference between variables but not the direction, nature, or magnitude of the relationship. A non-directional hypothesis may be used when there is no underlying theory or when findings contradict previous research.
Example, “ Cats and dogs differ in the amount of affection they express .”
5. Simple hypothesis :
A simple hypothesis only predicts the relationship between one independent and another independent variable.
Example: “ Applying sunscreen every day slows skin aging .”
6 . Complex hypothesis :
A complex hypothesis states the relationship or difference between two or more independent and dependent variables.
Example: “ Applying sunscreen every day slows skin aging, reduces sun burn, and reduces the chances of skin cancer .” (Here, the three dependent variables are slowing skin aging, reducing sun burn, and reducing the chances of skin cancer.)
7. Associative hypothesis:
An associative hypothesis states that a change in one variable results in the change of the other variable. The associative hypothesis defines interdependency between variables.
Example: “ There is a positive association between physical activity levels and overall health .”
8 . Causal hypothesis:
A causal hypothesis proposes a cause-and-effect interaction between variables.
Example: “ Long-term alcohol use causes liver damage .”
Note that some of the types of research hypothesis mentioned above might overlap. The types of hypothesis chosen will depend on the research question and the objective of the study.

Research hypothesis examples
Here are some good research hypothesis examples :
“The use of a specific type of therapy will lead to a reduction in symptoms of depression in individuals with a history of major depressive disorder.”
“Providing educational interventions on healthy eating habits will result in weight loss in overweight individuals.”
“Plants that are exposed to certain types of music will grow taller than those that are not exposed to music.”
“The use of the plant growth regulator X will lead to an increase in the number of flowers produced by plants.”
Characteristics that make a research hypothesis weak are unclear variables, unoriginality, being too general or too vague, and being untestable. A weak hypothesis leads to weak research and improper methods.
Some bad research hypothesis examples (and the reasons why they are “bad”) are as follows:
“This study will show that treatment X is better than any other treatment . ” (This statement is not testable, too broad, and does not consider other treatments that may be effective.)
“This study will prove that this type of therapy is effective for all mental disorders . ” (This statement is too broad and not testable as mental disorders are complex and different disorders may respond differently to different types of therapy.)
“Plants can communicate with each other through telepathy . ” (This statement is not testable and lacks a scientific basis.)
Importance of testable hypothesis
If a research hypothesis is not testable, the results will not prove or disprove anything meaningful. The conclusions will be vague at best. A testable hypothesis helps a researcher focus on the study outcome and understand the implication of the question and the different variables involved. A testable hypothesis helps a researcher make precise predictions based on prior research.
To be considered testable, there must be a way to prove that the hypothesis is true or false; further, the results of the hypothesis must be reproducible.

Frequently Asked Questions (FAQs) on research hypothesis
1. What is the difference between research question and research hypothesis ?
A research question defines the problem and helps outline the study objective(s). It is an open-ended statement that is exploratory or probing in nature. Therefore, it does not make predictions or assumptions. It helps a researcher identify what information to collect. A research hypothesis , however, is a specific, testable prediction about the relationship between variables. Accordingly, it guides the study design and data analysis approach.
2. When to reject null hypothesis ?
A null hypothesis should be rejected when the evidence from a statistical test shows that it is unlikely to be true. This happens when the test statistic (e.g., p -value) is less than the defined significance level (e.g., 0.05). Rejecting the null hypothesis does not necessarily mean that the alternative hypothesis is true; it simply means that the evidence found is not compatible with the null hypothesis.
3. How can I be sure my hypothesis is testable?
A testable hypothesis should be specific and measurable, and it should state a clear relationship between variables that can be tested with data. To ensure that your hypothesis is testable, consider the following:
- Clearly define the key variables in your hypothesis. You should be able to measure and manipulate these variables in a way that allows you to test the hypothesis.
- The hypothesis should predict a specific outcome or relationship between variables that can be measured or quantified.
- You should be able to collect the necessary data within the constraints of your study.
- It should be possible for other researchers to replicate your study, using the same methods and variables.
- Your hypothesis should be testable by using appropriate statistical analysis techniques, so you can draw conclusions, and make inferences about the population from the sample data.
- The hypothesis should be able to be disproven or rejected through the collection of data.
4. How do I revise my research hypothesis if my data does not support it?
If your data does not support your research hypothesis , you will need to revise it or develop a new one. You should examine your data carefully and identify any patterns or anomalies, re-examine your research question, and/or revisit your theory to look for any alternative explanations for your results. Based on your review of the data, literature, and theories, modify your research hypothesis to better align it with the results you obtained. Use your revised hypothesis to guide your research design and data collection. It is important to remain objective throughout the process.
5. I am performing exploratory research. Do I need to formulate a research hypothesis?
As opposed to “confirmatory” research, where a researcher has some idea about the relationship between the variables under investigation, exploratory research (or hypothesis-generating research) looks into a completely new topic about which limited information is available. Therefore, the researcher will not have any prior hypotheses. In such cases, a researcher will need to develop a post-hoc hypothesis. A post-hoc research hypothesis is generated after these results are known.
6. How is a research hypothesis different from a research question?
A research question is an inquiry about a specific topic or phenomenon, typically expressed as a question. It seeks to explore and understand a particular aspect of the research subject. In contrast, a research hypothesis is a specific statement or prediction that suggests an expected relationship between variables. It is formulated based on existing knowledge or theories and guides the research design and data analysis.
7. Can a research hypothesis change during the research process?
Yes, research hypotheses can change during the research process. As researchers collect and analyze data, new insights and information may emerge that require modification or refinement of the initial hypotheses. This can be due to unexpected findings, limitations in the original hypotheses, or the need to explore additional dimensions of the research topic. Flexibility is crucial in research, allowing for adaptation and adjustment of hypotheses to align with the evolving understanding of the subject matter.
8. How many hypotheses should be included in a research study?
The number of research hypotheses in a research study varies depending on the nature and scope of the research. It is not necessary to have multiple hypotheses in every study. Some studies may have only one primary hypothesis, while others may have several related hypotheses. The number of hypotheses should be determined based on the research objectives, research questions, and the complexity of the research topic. It is important to ensure that the hypotheses are focused, testable, and directly related to the research aims.
9. Can research hypotheses be used in qualitative research?
Yes, research hypotheses can be used in qualitative research, although they are more commonly associated with quantitative research. In qualitative research, hypotheses may be formulated as tentative or exploratory statements that guide the investigation. Instead of testing hypotheses through statistical analysis, qualitative researchers may use the hypotheses to guide data collection and analysis, seeking to uncover patterns, themes, or relationships within the qualitative data. The emphasis in qualitative research is often on generating insights and understanding rather than confirming or rejecting specific research hypotheses through statistical testing.
Editage All Access is a subscription-based platform that unifies the best AI tools and services designed to speed up, simplify, and streamline every step of a researcher’s journey. The Editage All Access Pack is a one-of-a-kind subscription that unlocks full access to an AI writing assistant, literature recommender, journal finder, scientific illustration tool, and exclusive discounts on professional publication services from Editage.
Based on 22+ years of experience in academia, Editage All Access empowers researchers to put their best research forward and move closer to success. Explore our top AI Tools pack, AI Tools + Publication Services pack, or Build Your Own Plan. Find everything a researcher needs to succeed, all in one place – Get All Access now starting at just $14 a month !
Related Posts

Back to School – Lock-in All Access Pack for a Year at the Best Price

Journal Turnaround Time: Researcher.Life and Scholarly Intelligence Join Hands to Empower Researchers with Publication Time Insights

Handy Tips To Write A Clear Research Objectives With Examples
Introduction.
Research objectives play a crucial role in any research study. They provide a clear direction and purpose for the research, guiding the researcher in their investigation. Understanding research objectives is essential for conducting a successful study and achieving meaningful results.
In this comprehensive review, we will delve into the definition of research objectives, exploring their characteristics, types, and examples. We will also discuss the relationship between research objectives and research questions , as well as provide insights into how to write effective research objectives. Additionally, we will examine the role of research objectives in research methodology and highlight the importance of them in a study. By the end of this review, you will have a comprehensive understanding of research objectives and their significance in the research process.
Definition of Research Objectives: What Are They?

A research objective is defined as a clear and concise statement that outlines the specific goals and aims of a research study. These objectives are designed to be specific, measurable, achievable, relevant, and time-bound (SMART), ensuring they provide a structured pathway to accomplishing the intended outcomes of the project. Each objective serves as a foundational element that summarizes the purpose of your study, guiding the research activities and helping to measure progress toward the study’s goals. Additionally, research objectives are integral components of the research framework , establishing a clear direction that aligns with the overall research questions and hypotheses. This alignment helps to ensure that the study remains focused and relevant, facilitating the systematic collection, analysis, and interpretation of data.
Characteristics of Effective Research Objectives
Characteristics of research objectives include:
- Specific: Research objectives should be clear about the what, why, when, and how of the study.
- Measurable: Research objectives should identify the main variables of the study that can be measured or observed.
- Relevant: Research objectives should be relevant to the research topic and contribute to the overall understanding of the subject.
- Feasible: Research objectives should be achievable within the constraints of time, resources, and expertise available.
- Logical: Research objectives should follow a logical sequence and build upon each other to achieve the overall research goal.
- Observable: Research objectives should be observable or measurable in order to assess the progress and success of the research project.
- Unambiguous: Research objectives should be clear and unambiguous, leaving no room for interpretation or confusion.
- Measurable: Research objectives should be measurable, allowing for the collection of data and analysis of results.
By incorporating these characteristics into research objectives, researchers can ensure that their study is focused, achievable, and contributes to the body of knowledge in their field.
Types of Research Objectives
Research objective can be broadly classified into general and specific objectives. General objectives are broad statements that define the overall purpose of the research. They provide a broad direction for the study and help in setting the context. Specific objectives, on the other hand, are detailed objectives that describe what will be researched during the study. They are more focused and provide specific outcomes that the researcher aims to achieve. Specific objectives are derived from the general objectives and help in breaking down the research into smaller, manageable parts. The specific objectives should be clear, measurable, and achievable. They should be designed in a way that allows the researcher to answer the research questions and address the research problem.
In addition to general and specific objectives, research objective can also be categorized as descriptive or analytical objectives. Descriptive objectives focus on describing the characteristics or phenomena of a particular subject or population. They involve surveys, observations, and data collection to provide a detailed understanding of the subject. Analytical objectives, on the other hand, aim to analyze the relationships between variables or factors. They involve data analysis and interpretation to gain insights and draw conclusions.
Both descriptive and analytical objectives are important in research as they serve different purposes and contribute to a comprehensive understanding of the research topic.
Examples of Research Objectives
Here are some examples of research objectives in different fields:
1. Objective: To identify key characteristics and styles of Renaissance art.
This objective focuses on exploring the characteristics and styles of art during the Renaissance period. The research may involve analyzing various artworks, studying historical documents, and interviewing experts in the field.
2. Objective: To analyze modern art trends and their impact on society.
This objective aims to examine the current trends in modern art and understand how they influence society. The research may involve analyzing artworks, conducting surveys or interviews with artists and art enthusiasts, and studying the social and cultural implications of modern art.
3. Objective: To investigate the effects of exercise on mental health.
This objective focuses on studying the relationship between exercise and mental health. The research may involve conducting experiments or surveys to assess the impact of exercise on factors such as stress, anxiety, and depression.
4. Objective: To explore the factors influencing consumer purchasing decisions in the fashion industry.
This objective aims to understand the various factors that influence consumers’ purchasing decisions in the fashion industry. The research may involve conducting surveys, analyzing consumer behavior data, and studying the impact of marketing strategies on consumer choices.
5. Objective: To examine the effectiveness of a new drug in treating a specific medical condition.
This objective focuses on evaluating the effectiveness of a newly developed drug in treating a particular medical condition. The research may involve conducting clinical trials, analyzing patient data, and comparing the outcomes of the new drug with existing treatment options.
These examples demonstrate the diversity of research objectives across different disciplines. Each objective is specific, measurable, and achievable, providing a clear direction for the research study.
Aligning Research Objectives with Research Questions
Research objectives and research questions are essential components of a research project. Research objective describe what you intend your research project to accomplish. They summarize the approach and purpose of the project and provide a clear direction for the research. Research questions, on the other hand, are the starting point of any good research. They guide the overall direction of the research and help identify and focus on the research gaps .
The main difference between research questions and objectives is their form. Research questions are stated in a question form, while objectives are specific, measurable, and achievable goals that you aim to accomplish within a specified timeframe. Research questions are broad statements that provide a roadmap for the research, while objectives break down the research aim into smaller, actionable steps.
Research objectives and research questions work together to form the ‘golden thread’ of a research project. The research aim specifies what the study will answer, while the objectives and questions specify how the study will answer it. They provide a clear focus and scope for the research project, helping researchers stay on track and ensure that their study is meaningful and relevant.
When writing research objectives and questions, it is important to be clear, concise, and specific. Each objective or question should address a specific aspect of the research and contribute to the overall goal of the study. They should also be measurable, meaning that their achievement can be assessed and evaluated. Additionally, research objectives and questions should be achievable within the given timeframe and resources of the research project. By clearly defining the objectives and questions, researchers can effectively plan and execute their research, leading to valuable insights and contributions to the field.
Guidelines for Writing Clear Research Objectives
Writing research objective is a crucial step in any research project. The objectives provide a clear direction and purpose for the study, guiding the researcher in their data collection and analysis. Here are some tips on how to write effective research objective:
1. Be clear and specific
Research objective should be written in a clear and specific manner. Avoid vague or ambiguous language that can lead to confusion. Clearly state what you intend to achieve through your research.
2. Use action verbs
Start your research objective with action verbs that describe the desired outcome. Action verbs such as ‘investigate’, ‘analyze’, ‘compare’, ‘evaluate’, or ‘identify’ help to convey the purpose of the study.
3. Align with research questions or hypotheses
Ensure that your research objectives are aligned with your research questions or hypotheses. The objectives should address the main goals of your study and provide a framework for answering your research questions or testing your hypotheses.
4. Be realistic and achievable
Set research objectives that are realistic and achievable within the scope of your study. Consider the available resources, time constraints, and feasibility of your objectives. Unrealistic objectives can lead to frustration and hinder the progress of your research.
5. Consider the significance and relevance
Reflect on the significance and relevance of your research objectives. How will achieving these objectives contribute to the existing knowledge or address a gap in the literature? Ensure that your objectives have a clear purpose and value.
6. Seek feedback
It is beneficial to seek feedback on your research objectives from colleagues, mentors, or experts in your field. They can provide valuable insights and suggestions for improving the clarity and effectiveness of your objectives.
7. Revise and refine
Research objectives are not set in stone. As you progress in your research, you may need to revise and refine your objectives to align with new findings or changes in the research context. Regularly review and update your objectives to ensure they remain relevant and focused.
By following these tips, you can write research objectives that are clear, focused, and aligned with your research goals. Well-defined objectives will guide your research process and help you achieve meaningful outcomes.
The Role of Research Objectives in Research Methodology
Research objectives play a crucial role in the research methodology . In research methodology, research objectives are formulated based on the research questions or problem statement. These objectives help in defining the scope and focus of the study, ensuring that the research is conducted in a systematic and organized manner.
The research objectives in research methodology act as a roadmap for the research project. They help in identifying the key variables to be studied, determining the research design and methodology, and selecting the appropriate data collection methods .
Furthermore, research objectives in research methodology assist in evaluating the success of the study. By setting clear objectives, researchers can assess whether the desired outcomes have been achieved and determine the effectiveness of the research methods employed. It is important to note that research objectives in research methodology should be aligned with the overall research aim. They should address the specific aspects or components of the research aim and provide a framework for achieving the desired outcomes.
Understanding The Dynamic of Research Objectives in Your Study
The research objectives of a study play a crucial role in guiding the research process, ensuring that the study is focused, purposeful, and contributes to the advancement of knowledge in the field. It is important to note that the research objectives may evolve or change as the study progresses. As new information is gathered and analyzed, the researcher may need to revise the objectives to ensure that they remain relevant and achievable.
In summary, research objectives are essential components in writing an effective research paper . They provide a roadmap for the research process, guiding the researcher in their investigation and helping to ensure that the study is purposeful and meaningful. By understanding and effectively utilizing research objectives, researchers can enhance the quality and impact of their research endeavors.
Leave a Comment Cancel reply
Save my name, email, and website in this browser for the next time I comment.
Related articles

How to Formulate Research Questions in a Research Proposal? Discover The No. 1 Easiest Template Here!

7 Easy Step-By-Step Guide of Using ChatGPT: The Best Literature Review Generator for Time-Saving Academic Research

Writing Engaging Introduction in Research Papers : 7 Tips and Tricks!

Understanding Comparative Frameworks: Their Importance, Components, Examples and 8 Best Practices
Revolutionizing Effective Thesis Writing for PhD Students Using Artificial Intelligence!

3 Types of Interviews in Qualitative Research: An Essential Research Instrument and Handy Tips to Conduct Them

Highlight Abstracts: An Ultimate Guide For Researchers!

Crafting Critical Abstracts: 11 Expert Strategies for Summarizing Research
- Skip to main content
- Skip to primary sidebar
- Skip to footer
- QuestionPro

- Solutions Industries Gaming Automotive Sports and events Education Government Travel & Hospitality Financial Services Healthcare Cannabis Technology Use Case AskWhy Communities Audience Contactless surveys Mobile LivePolls Member Experience GDPR Positive People Science 360 Feedback Surveys
- Resources Blog eBooks Survey Templates Case Studies Training Help center
Home Market Research
Research Hypothesis: What It Is, Types + How to Develop?

A research study starts with a question. Researchers worldwide ask questions and create research hypotheses. The effectiveness of research relies on developing a good research hypothesis. Examples of research hypotheses can guide researchers in writing effective ones.
In this blog, we’ll learn what a research hypothesis is, why it’s important in research, and the different types used in science. We’ll also guide you through creating your research hypothesis and discussing ways to test and evaluate it.
What is a Research Hypothesis?
A hypothesis is like a guess or idea that you suggest to check if it’s true. A research hypothesis is a statement that brings up a question and predicts what might happen.
It’s really important in the scientific method and is used in experiments to figure things out. Essentially, it’s an educated guess about how things are connected in the research.
A research hypothesis usually includes pointing out the independent variable (the thing they’re changing or studying) and the dependent variable (the result they’re measuring or watching). It helps plan how to gather and analyze data to see if there’s evidence to support or deny the expected connection between these variables.
Importance of Hypothesis in Research
Hypotheses are really important in research. They help design studies, allow for practical testing, and add to our scientific knowledge. Their main role is to organize research projects, making them purposeful, focused, and valuable to the scientific community. Let’s look at some key reasons why they matter:
- A research hypothesis helps test theories.
A hypothesis plays a pivotal role in the scientific method by providing a basis for testing existing theories. For example, a hypothesis might test the predictive power of a psychological theory on human behavior.
- It serves as a great platform for investigation activities.
It serves as a launching pad for investigation activities, which offers researchers a clear starting point. A research hypothesis can explore the relationship between exercise and stress reduction.
- Hypothesis guides the research work or study.
A well-formulated hypothesis guides the entire research process. It ensures that the study remains focused and purposeful. For instance, a hypothesis about the impact of social media on interpersonal relationships provides clear guidance for a study.
- Hypothesis sometimes suggests theories.
In some cases, a hypothesis can suggest new theories or modifications to existing ones. For example, a hypothesis testing the effectiveness of a new drug might prompt a reconsideration of current medical theories.
- It helps in knowing the data needs.
A hypothesis clarifies the data requirements for a study, ensuring that researchers collect the necessary information—a hypothesis guiding the collection of demographic data to analyze the influence of age on a particular phenomenon.
- The hypothesis explains social phenomena.
Hypotheses are instrumental in explaining complex social phenomena. For instance, a hypothesis might explore the relationship between economic factors and crime rates in a given community.
- Hypothesis provides a relationship between phenomena for empirical Testing.
Hypotheses establish clear relationships between phenomena, paving the way for empirical testing. An example could be a hypothesis exploring the correlation between sleep patterns and academic performance.
- It helps in knowing the most suitable analysis technique.
A hypothesis guides researchers in selecting the most appropriate analysis techniques for their data. For example, a hypothesis focusing on the effectiveness of a teaching method may lead to the choice of statistical analyses best suited for educational research.
Characteristics of a Good Research Hypothesis
A hypothesis is a specific idea that you can test in a study. It often comes from looking at past research and theories. A good hypothesis usually starts with a research question that you can explore through background research. For it to be effective, consider these key characteristics:
- Clear and Focused Language: A good hypothesis uses clear and focused language to avoid confusion and ensure everyone understands it.
- Related to the Research Topic: The hypothesis should directly relate to the research topic, acting as a bridge between the specific question and the broader study.
- Testable: An effective hypothesis can be tested, meaning its prediction can be checked with real data to support or challenge the proposed relationship.
- Potential for Exploration: A good hypothesis often comes from a research question that invites further exploration. Doing background research helps find gaps and potential areas to investigate.
- Includes Variables: The hypothesis should clearly state both the independent and dependent variables, specifying the factors being studied and the expected outcomes.
- Ethical Considerations: Check if variables can be manipulated without breaking ethical standards. It’s crucial to maintain ethical research practices.
- Predicts Outcomes: The hypothesis should predict the expected relationship and outcome, acting as a roadmap for the study and guiding data collection and analysis.
- Simple and Concise: A good hypothesis avoids unnecessary complexity and is simple and concise, expressing the essence of the proposed relationship clearly.
- Clear and Assumption-Free: The hypothesis should be clear and free from assumptions about the reader’s prior knowledge, ensuring universal understanding.
- Observable and Testable Results: A strong hypothesis implies research that produces observable and testable results, making sure the study’s outcomes can be effectively measured and analyzed.
When you use these characteristics as a checklist, it can help you create a good research hypothesis. It’ll guide improving and strengthening the hypothesis, identifying any weaknesses, and making necessary changes. Crafting a hypothesis with these features helps you conduct a thorough and insightful research study.
Types of Research Hypotheses
The research hypothesis comes in various types, each serving a specific purpose in guiding the scientific investigation. Knowing the differences will make it easier for you to create your own hypothesis. Here’s an overview of the common types:

01. Null Hypothesis
The null hypothesis states that there is no connection between two considered variables or that two groups are unrelated. As discussed earlier, a hypothesis is an unproven assumption lacking sufficient supporting data. It serves as the statement researchers aim to disprove. It is testable, verifiable, and can be rejected.
For example, if you’re studying the relationship between Project A and Project B, assuming both projects are of equal standard is your null hypothesis. It needs to be specific for your study.
02. Alternative Hypothesis
The alternative hypothesis is basically another option to the null hypothesis. It involves looking for a significant change or alternative that could lead you to reject the null hypothesis. It’s a different idea compared to the null hypothesis.
When you create a null hypothesis, you’re making an educated guess about whether something is true or if there’s a connection between that thing and another variable. If the null view suggests something is correct, the alternative hypothesis says it’s incorrect.
For instance, if your null hypothesis is “I’m going to be $1000 richer,” the alternative hypothesis would be “I’m not going to get $1000 or be richer.”
03. Directional Hypothesis
The directional hypothesis predicts the direction of the relationship between independent and dependent variables. They specify whether the effect will be positive or negative.
If you increase your study hours, you will experience a positive association with your exam scores. This hypothesis suggests that as you increase the independent variable (study hours), there will also be an increase in the dependent variable (exam scores).
04. Non-directional Hypothesis
The non-directional hypothesis predicts the existence of a relationship between variables but does not specify the direction of the effect. It suggests that there will be a significant difference or relationship, but it does not predict the nature of that difference.
For example, you will find no notable difference in test scores between students who receive the educational intervention and those who do not. However, once you compare the test scores of the two groups, you will notice an important difference.
05. Simple Hypothesis
A simple hypothesis predicts a relationship between one dependent variable and one independent variable without specifying the nature of that relationship. It’s simple and usually used when we don’t know much about how the two things are connected.
For example, if you adopt effective study habits, you will achieve higher exam scores than those with poor study habits.
06. Complex Hypothesis
A complex hypothesis is an idea that specifies a relationship between multiple independent and dependent variables. It is a more detailed idea than a simple hypothesis.
While a simple view suggests a straightforward cause-and-effect relationship between two things, a complex hypothesis involves many factors and how they’re connected to each other.
For example, when you increase your study time, you tend to achieve higher exam scores. The connection between your study time and exam performance is affected by various factors, including the quality of your sleep, your motivation levels, and the effectiveness of your study techniques.
If you sleep well, stay highly motivated, and use effective study strategies, you may observe a more robust positive correlation between the time you spend studying and your exam scores, unlike those who may lack these factors.
07. Associative Hypothesis
An associative hypothesis proposes a connection between two things without saying that one causes the other. Basically, it suggests that when one thing changes, the other changes too, but it doesn’t claim that one thing is causing the change in the other.
For example, you will likely notice higher exam scores when you increase your study time. You can recognize an association between your study time and exam scores in this scenario.
Your hypothesis acknowledges a relationship between the two variables—your study time and exam scores—without asserting that increased study time directly causes higher exam scores. You need to consider that other factors, like motivation or learning style, could affect the observed association.
08. Causal Hypothesis
A causal hypothesis proposes a cause-and-effect relationship between two variables. It suggests that changes in one variable directly cause changes in another variable.
For example, when you increase your study time, you experience higher exam scores. This hypothesis suggests a direct cause-and-effect relationship, indicating that the more time you spend studying, the higher your exam scores. It assumes that changes in your study time directly influence changes in your exam performance.
09. Empirical Hypothesis
An empirical hypothesis is a statement based on things we can see and measure. It comes from direct observation or experiments and can be tested with real-world evidence. If an experiment proves a theory, it supports the idea and shows it’s not just a guess. This makes the statement more reliable than a wild guess.
For example, if you increase the dosage of a certain medication, you might observe a quicker recovery time for patients. Imagine you’re in charge of a clinical trial. In this trial, patients are given varying dosages of the medication, and you measure and compare their recovery times. This allows you to directly see the effects of different dosages on how fast patients recover.
This way, you can create a research hypothesis: “Increasing the dosage of a certain medication will lead to a faster recovery time for patients.”
10. Statistical Hypothesis
A statistical hypothesis is a statement or assumption about a population parameter that is the subject of an investigation. It serves as the basis for statistical analysis and testing. It is often tested using statistical methods to draw inferences about the larger population.
In a hypothesis test, statistical evidence is collected to either reject the null hypothesis in favor of the alternative hypothesis or fail to reject the null hypothesis due to insufficient evidence.
For example, let’s say you’re testing a new medicine. Your hypothesis could be that the medicine doesn’t really help patients get better. So, you collect data and use statistics to see if your guess is right or if the medicine actually makes a difference.
If the data strongly shows that the medicine does help, you say your guess was wrong, and the medicine does make a difference. But if the proof isn’t strong enough, you can stick with your original guess because you didn’t get enough evidence to change your mind.
How to Develop a Research Hypotheses?
Step 1: identify your research problem or topic..
Define the area of interest or the problem you want to investigate. Make sure it’s clear and well-defined.
Start by asking a question about your chosen topic. Consider the limitations of your research and create a straightforward problem related to your topic. Once you’ve done that, you can develop and test a hypothesis with evidence.
Step 2: Conduct a literature review
Review existing literature related to your research problem. This will help you understand the current state of knowledge in the field, identify gaps, and build a foundation for your hypothesis. Consider the following questions:
- What existing research has been conducted on your chosen topic?
- Are there any gaps or unanswered questions in the current literature?
- How will the existing literature contribute to the foundation of your research?
Step 3: Formulate your research question
Based on your literature review, create a specific and concise research question that addresses your identified problem. Your research question should be clear, focused, and relevant to your field of study.
Step 4: Identify variables
Determine the key variables involved in your research question. Variables are the factors or phenomena that you will study and manipulate to test your hypothesis.
- Independent Variable: The variable you manipulate or control.
- Dependent Variable: The variable you measure to observe the effect of the independent variable.
Step 5: State the Null hypothesis
The null hypothesis is a statement that there is no significant difference or effect. It serves as a baseline for comparison with the alternative hypothesis.
Step 6: Select appropriate methods for testing the hypothesis
Choose research methods that align with your study objectives, such as experiments, surveys, or observational studies. The selected methods enable you to test your research hypothesis effectively.
Creating a research hypothesis usually takes more than one try. Expect to make changes as you collect data. It’s normal to test and say no to a few hypotheses before you find the right answer to your research question.
Testing and Evaluating Hypotheses
Testing hypotheses is a really important part of research. It’s like the practical side of things. Here, real-world evidence will help you determine how different things are connected. Let’s explore the main steps in hypothesis testing:
- State your research hypothesis.
Before testing, clearly articulate your research hypothesis. This involves framing both a null hypothesis, suggesting no significant effect or relationship, and an alternative hypothesis, proposing the expected outcome.
- Collect data strategically.
Plan how you will gather information in a way that fits your study. Make sure your data collection method matches the things you’re studying.
Whether through surveys, observations, or experiments, this step demands precision and adherence to the established methodology. The quality of data collected directly influences the credibility of study outcomes.
- Perform an appropriate statistical test.
Choose a statistical test that aligns with the nature of your data and the hypotheses being tested. Whether it’s a t-test, chi-square test, ANOVA, or regression analysis, selecting the right statistical tool is paramount for accurate and reliable results.
- Decide if your idea was right or wrong.
Following the statistical analysis, evaluate the results in the context of your null hypothesis. You need to decide if you should reject your null hypothesis or not.
- Share what you found.
When discussing what you found in your research, be clear and organized. Say whether your idea was supported or not, and talk about what your results mean. Also, mention any limits to your study and suggest ideas for future research.
The Role of QuestionPro to Develop a Good Research Hypothesis
QuestionPro is a survey and research platform that provides tools for creating, distributing, and analyzing surveys. It plays a crucial role in the research process, especially when you’re in the initial stages of hypothesis development. Here’s how QuestionPro can help you to develop a good research hypothesis:
- Survey design and data collection: You can use the platform to create targeted questions that help you gather relevant data.
- Exploratory research: Through surveys and feedback mechanisms on QuestionPro, you can conduct exploratory research to understand the landscape of a particular subject.
- Literature review and background research: QuestionPro surveys can collect sample population opinions, experiences, and preferences. This data and a thorough literature evaluation can help you generate a well-grounded hypothesis by improving your research knowledge.
- Identifying variables: Using targeted survey questions, you can identify relevant variables related to their research topic.
- Testing assumptions: You can use surveys to informally test certain assumptions or hypotheses before formalizing a research hypothesis.
- Data analysis tools: QuestionPro provides tools for analyzing survey data. You can use these tools to identify the collected data’s patterns, correlations, or trends.
- Refining your hypotheses: As you collect data through QuestionPro, you can adjust your hypotheses based on the real-world responses you receive.
A research hypothesis is like a guide for researchers in science. It’s a well-thought-out idea that has been thoroughly tested. This idea is crucial as researchers can explore different fields, such as medicine, social sciences, and natural sciences. The research hypothesis links theories to real-world evidence and gives researchers a clear path to explore and make discoveries.
QuestionPro Research Suite is a helpful tool for researchers. It makes creating surveys, collecting data, and analyzing information easily. It supports all kinds of research, from exploring new ideas to forming hypotheses. With a focus on using data, it helps researchers do their best work.
Are you interested in learning more about QuestionPro Research Suite? Take advantage of QuestionPro’s free trial to get an initial look at its capabilities and realize the full potential of your research efforts.
LEARN MORE FREE TRIAL
MORE LIKE THIS

Interactive Forms: Key Features, Benefits, Uses + Design Tips
Sep 4, 2024

Closed-Loop Management: The Key to Customer Centricity
Sep 3, 2024

Net Trust Score: Tool for Measuring Trust in Organization
Sep 2, 2024

Why You Should Attend XDAY 2024
Aug 30, 2024
Other categories
- Academic Research
- Artificial Intelligence
- Assessments
- Brand Awareness
- Case Studies
- Communities
- Consumer Insights
- Customer effort score
- Customer Engagement
- Customer Experience
- Customer Loyalty
- Customer Research
- Customer Satisfaction
- Employee Benefits
- Employee Engagement
- Employee Retention
- Friday Five
- General Data Protection Regulation
- Insights Hub
- Life@QuestionPro
- Market Research
- Mobile diaries
- Mobile Surveys
- New Features
- Online Communities
- Question Types
- Questionnaire
- QuestionPro Products
- Release Notes
- Research Tools and Apps
- Revenue at Risk
- Survey Templates
- Training Tips
- Tuesday CX Thoughts (TCXT)
- Uncategorized
- What’s Coming Up
- Workforce Intelligence

What Is A Research (Scientific) Hypothesis? A plain-language explainer + examples
By: Derek Jansen (MBA) | Reviewed By: Dr Eunice Rautenbach | June 2020
If you’re new to the world of research, or it’s your first time writing a dissertation or thesis, you’re probably noticing that the words “research hypothesis” and “scientific hypothesis” are used quite a bit, and you’re wondering what they mean in a research context .
“Hypothesis” is one of those words that people use loosely, thinking they understand what it means. However, it has a very specific meaning within academic research. So, it’s important to understand the exact meaning before you start hypothesizing.
Research Hypothesis 101
- What is a hypothesis ?
- What is a research hypothesis (scientific hypothesis)?
- Requirements for a research hypothesis
- Definition of a research hypothesis
- The null hypothesis
What is a hypothesis?
Let’s start with the general definition of a hypothesis (not a research hypothesis or scientific hypothesis), according to the Cambridge Dictionary:
Hypothesis: an idea or explanation for something that is based on known facts but has not yet been proved.
In other words, it’s a statement that provides an explanation for why or how something works, based on facts (or some reasonable assumptions), but that has not yet been specifically tested . For example, a hypothesis might look something like this:
Hypothesis: sleep impacts academic performance.
This statement predicts that academic performance will be influenced by the amount and/or quality of sleep a student engages in – sounds reasonable, right? It’s based on reasonable assumptions , underpinned by what we currently know about sleep and health (from the existing literature). So, loosely speaking, we could call it a hypothesis, at least by the dictionary definition.
But that’s not good enough…
Unfortunately, that’s not quite sophisticated enough to describe a research hypothesis (also sometimes called a scientific hypothesis), and it wouldn’t be acceptable in a dissertation, thesis or research paper . In the world of academic research, a statement needs a few more criteria to constitute a true research hypothesis .
What is a research hypothesis?
A research hypothesis (also called a scientific hypothesis) is a statement about the expected outcome of a study (for example, a dissertation or thesis). To constitute a quality hypothesis, the statement needs to have three attributes – specificity , clarity and testability .
Let’s take a look at these more closely.
Need a helping hand?
Hypothesis Essential #1: Specificity & Clarity
A good research hypothesis needs to be extremely clear and articulate about both what’ s being assessed (who or what variables are involved ) and the expected outcome (for example, a difference between groups, a relationship between variables, etc.).
Let’s stick with our sleepy students example and look at how this statement could be more specific and clear.
Hypothesis: Students who sleep at least 8 hours per night will, on average, achieve higher grades in standardised tests than students who sleep less than 8 hours a night.
As you can see, the statement is very specific as it identifies the variables involved (sleep hours and test grades), the parties involved (two groups of students), as well as the predicted relationship type (a positive relationship). There’s no ambiguity or uncertainty about who or what is involved in the statement, and the expected outcome is clear.
Contrast that to the original hypothesis we looked at – “Sleep impacts academic performance” – and you can see the difference. “Sleep” and “academic performance” are both comparatively vague , and there’s no indication of what the expected relationship direction is (more sleep or less sleep). As you can see, specificity and clarity are key.

Hypothesis Essential #2: Testability (Provability)
A statement must be testable to qualify as a research hypothesis. In other words, there needs to be a way to prove (or disprove) the statement. If it’s not testable, it’s not a hypothesis – simple as that.
For example, consider the hypothesis we mentioned earlier:
Hypothesis: Students who sleep at least 8 hours per night will, on average, achieve higher grades in standardised tests than students who sleep less than 8 hours a night.
We could test this statement by undertaking a quantitative study involving two groups of students, one that gets 8 or more hours of sleep per night for a fixed period, and one that gets less. We could then compare the standardised test results for both groups to see if there’s a statistically significant difference.
Again, if you compare this to the original hypothesis we looked at – “Sleep impacts academic performance” – you can see that it would be quite difficult to test that statement, primarily because it isn’t specific enough. How much sleep? By who? What type of academic performance?
So, remember the mantra – if you can’t test it, it’s not a hypothesis 🙂

Defining A Research Hypothesis
You’re still with us? Great! Let’s recap and pin down a clear definition of a hypothesis.
A research hypothesis (or scientific hypothesis) is a statement about an expected relationship between variables, or explanation of an occurrence, that is clear, specific and testable.
So, when you write up hypotheses for your dissertation or thesis, make sure that they meet all these criteria. If you do, you’ll not only have rock-solid hypotheses but you’ll also ensure a clear focus for your entire research project.
What about the null hypothesis?
You may have also heard the terms null hypothesis , alternative hypothesis, or H-zero thrown around. At a simple level, the null hypothesis is the counter-proposal to the original hypothesis.
For example, if the hypothesis predicts that there is a relationship between two variables (for example, sleep and academic performance), the null hypothesis would predict that there is no relationship between those variables.
At a more technical level, the null hypothesis proposes that no statistical significance exists in a set of given observations and that any differences are due to chance alone.
And there you have it – hypotheses in a nutshell.
If you have any questions, be sure to leave a comment below and we’ll do our best to help you. If you need hands-on help developing and testing your hypotheses, consider our private coaching service , where we hold your hand through the research journey.

17 Comments
Very useful information. I benefit more from getting more information in this regard.
Very great insight,educative and informative. Please give meet deep critics on many research data of public international Law like human rights, environment, natural resources, law of the sea etc
In a book I read a distinction is made between null, research, and alternative hypothesis. As far as I understand, alternative and research hypotheses are the same. Can you please elaborate? Best Afshin
This is a self explanatory, easy going site. I will recommend this to my friends and colleagues.
Very good definition. How can I cite your definition in my thesis? Thank you. Is nul hypothesis compulsory in a research?
It’s a counter-proposal to be proven as a rejection
Please what is the difference between alternate hypothesis and research hypothesis?
It is a very good explanation. However, it limits hypotheses to statistically tasteable ideas. What about for qualitative researches or other researches that involve quantitative data that don’t need statistical tests?
In qualitative research, one typically uses propositions, not hypotheses.
could you please elaborate it more
I’ve benefited greatly from these notes, thank you.
This is very helpful
well articulated ideas are presented here, thank you for being reliable sources of information
Excellent. Thanks for being clear and sound about the research methodology and hypothesis (quantitative research)
I have only a simple question regarding the null hypothesis. – Is the null hypothesis (Ho) known as the reversible hypothesis of the alternative hypothesis (H1? – How to test it in academic research?
this is very important note help me much more
Hi” best wishes to you and your very nice blog”
Trackbacks/Pingbacks
- What Is Research Methodology? Simple Definition (With Examples) - Grad Coach - […] Contrasted to this, a quantitative methodology is typically used when the research aims and objectives are confirmatory in nature. For example,…
Submit a Comment Cancel reply
Your email address will not be published. Required fields are marked *
Save my name, email, and website in this browser for the next time I comment.
- Print Friendly

- Aims and Objectives – A Guide for Academic Writing
- Doing a PhD
One of the most important aspects of a thesis, dissertation or research paper is the correct formulation of the aims and objectives. This is because your aims and objectives will establish the scope, depth and direction that your research will ultimately take. An effective set of aims and objectives will give your research focus and your reader clarity, with your aims indicating what is to be achieved, and your objectives indicating how it will be achieved.
Introduction
There is no getting away from the importance of the aims and objectives in determining the success of your research project. Unfortunately, however, it is an aspect that many students struggle with, and ultimately end up doing poorly. Given their importance, if you suspect that there is even the smallest possibility that you belong to this group of students, we strongly recommend you read this page in full.
This page describes what research aims and objectives are, how they differ from each other, how to write them correctly, and the common mistakes students make and how to avoid them. An example of a good aim and objectives from a past thesis has also been deconstructed to help your understanding.
What Are Aims and Objectives?
Research aims.
A research aim describes the main goal or the overarching purpose of your research project.
In doing so, it acts as a focal point for your research and provides your readers with clarity as to what your study is all about. Because of this, research aims are almost always located within its own subsection under the introduction section of a research document, regardless of whether it’s a thesis , a dissertation, or a research paper .
A research aim is usually formulated as a broad statement of the main goal of the research and can range in length from a single sentence to a short paragraph. Although the exact format may vary according to preference, they should all describe why your research is needed (i.e. the context), what it sets out to accomplish (the actual aim) and, briefly, how it intends to accomplish it (overview of your objectives).
To give an example, we have extracted the following research aim from a real PhD thesis:
Example of a Research Aim
The role of diametrical cup deformation as a factor to unsatisfactory implant performance has not been widely reported. The aim of this thesis was to gain an understanding of the diametrical deformation behaviour of acetabular cups and shells following impaction into the reamed acetabulum. The influence of a range of factors on deformation was investigated to ascertain if cup and shell deformation may be high enough to potentially contribute to early failure and high wear rates in metal-on-metal implants.
Note: Extracted with permission from thesis titled “T he Impact And Deformation Of Press-Fit Metal Acetabular Components ” produced by Dr H Hothi of previously Queen Mary University of London.
Research Objectives
Where a research aim specifies what your study will answer, research objectives specify how your study will answer it.
They divide your research aim into several smaller parts, each of which represents a key section of your research project. As a result, almost all research objectives take the form of a numbered list, with each item usually receiving its own chapter in a dissertation or thesis.
Following the example of the research aim shared above, here are it’s real research objectives as an example:
Example of a Research Objective
- Develop finite element models using explicit dynamics to mimic mallet blows during cup/shell insertion, initially using simplified experimentally validated foam models to represent the acetabulum.
- Investigate the number, velocity and position of impacts needed to insert a cup.
- Determine the relationship between the size of interference between the cup and cavity and deformation for different cup types.
- Investigate the influence of non-uniform cup support and varying the orientation of the component in the cavity on deformation.
- Examine the influence of errors during reaming of the acetabulum which introduce ovality to the cavity.
- Determine the relationship between changes in the geometry of the component and deformation for different cup designs.
- Develop three dimensional pelvis models with non-uniform bone material properties from a range of patients with varying bone quality.
- Use the key parameters that influence deformation, as identified in the foam models to determine the range of deformations that may occur clinically using the anatomic models and if these deformations are clinically significant.
It’s worth noting that researchers sometimes use research questions instead of research objectives, or in other cases both. From a high-level perspective, research questions and research objectives make the same statements, but just in different formats.
Taking the first three research objectives as an example, they can be restructured into research questions as follows:
Restructuring Research Objectives as Research Questions
- Can finite element models using simplified experimentally validated foam models to represent the acetabulum together with explicit dynamics be used to mimic mallet blows during cup/shell insertion?
- What is the number, velocity and position of impacts needed to insert a cup?
- What is the relationship between the size of interference between the cup and cavity and deformation for different cup types?
Difference Between Aims and Objectives
Hopefully the above explanations make clear the differences between aims and objectives, but to clarify:
- The research aim focus on what the research project is intended to achieve; research objectives focus on how the aim will be achieved.
- Research aims are relatively broad; research objectives are specific.
- Research aims focus on a project’s long-term outcomes; research objectives focus on its immediate, short-term outcomes.
- A research aim can be written in a single sentence or short paragraph; research objectives should be written as a numbered list.
How to Write Aims and Objectives
Before we discuss how to write a clear set of research aims and objectives, we should make it clear that there is no single way they must be written. Each researcher will approach their aims and objectives slightly differently, and often your supervisor will influence the formulation of yours on the basis of their own preferences.
Regardless, there are some basic principles that you should observe for good practice; these principles are described below.
Your aim should be made up of three parts that answer the below questions:
- Why is this research required?
- What is this research about?
- How are you going to do it?
The easiest way to achieve this would be to address each question in its own sentence, although it does not matter whether you combine them or write multiple sentences for each, the key is to address each one.
The first question, why , provides context to your research project, the second question, what , describes the aim of your research, and the last question, how , acts as an introduction to your objectives which will immediately follow.
Scroll through the image set below to see the ‘why, what and how’ associated with our research aim example.

Note: Your research aims need not be limited to one. Some individuals per to define one broad ‘overarching aim’ of a project and then adopt two or three specific research aims for their thesis or dissertation. Remember, however, that in order for your assessors to consider your research project complete, you will need to prove you have fulfilled all of the aims you set out to achieve. Therefore, while having more than one research aim is not necessarily disadvantageous, consider whether a single overarching one will do.
Research Objectives
Each of your research objectives should be SMART :
- Specific – is there any ambiguity in the action you are going to undertake, or is it focused and well-defined?
- Measurable – how will you measure progress and determine when you have achieved the action?
- Achievable – do you have the support, resources and facilities required to carry out the action?
- Relevant – is the action essential to the achievement of your research aim?
- Timebound – can you realistically complete the action in the available time alongside your other research tasks?
In addition to being SMART, your research objectives should start with a verb that helps communicate your intent. Common research verbs include:
Table of Research Verbs to Use in Aims and Objectives
| (Understanding and organising information) | (Solving problems using information) | (reaching conclusion from evidence) | (Breaking down into components) | (Judging merit) |
| Review Identify Explore Discover Discuss Summarise Describe | Interpret Apply Demonstrate Establish Determine Estimate Calculate Relate | Analyse Compare Inspect Examine Verify Select Test Arrange | Propose Design Formulate Collect Construct Prepare Undertake Assemble | Appraise Evaluate Compare Assess Recommend Conclude Select |
Last, format your objectives into a numbered list. This is because when you write your thesis or dissertation, you will at times need to make reference to a specific research objective; structuring your research objectives in a numbered list will provide a clear way of doing this.
To bring all this together, let’s compare the first research objective in the previous example with the above guidance:
Checking Research Objective Example Against Recommended Approach
Research Objective:
1. Develop finite element models using explicit dynamics to mimic mallet blows during cup/shell insertion, initially using simplified experimentally validated foam models to represent the acetabulum.
Checking Against Recommended Approach:
Q: Is it specific? A: Yes, it is clear what the student intends to do (produce a finite element model), why they intend to do it (mimic cup/shell blows) and their parameters have been well-defined ( using simplified experimentally validated foam models to represent the acetabulum ).
Q: Is it measurable? A: Yes, it is clear that the research objective will be achieved once the finite element model is complete.
Q: Is it achievable? A: Yes, provided the student has access to a computer lab, modelling software and laboratory data.
Q: Is it relevant? A: Yes, mimicking impacts to a cup/shell is fundamental to the overall aim of understanding how they deform when impacted upon.
Q: Is it timebound? A: Yes, it is possible to create a limited-scope finite element model in a relatively short time, especially if you already have experience in modelling.
Q: Does it start with a verb? A: Yes, it starts with ‘develop’, which makes the intent of the objective immediately clear.
Q: Is it a numbered list? A: Yes, it is the first research objective in a list of eight.
Mistakes in Writing Research Aims and Objectives
1. making your research aim too broad.
Having a research aim too broad becomes very difficult to achieve. Normally, this occurs when a student develops their research aim before they have a good understanding of what they want to research. Remember that at the end of your project and during your viva defence , you will have to prove that you have achieved your research aims; if they are too broad, this will be an almost impossible task. In the early stages of your research project, your priority should be to narrow your study to a specific area. A good way to do this is to take the time to study existing literature, question their current approaches, findings and limitations, and consider whether there are any recurring gaps that could be investigated .
Note: Achieving a set of aims does not necessarily mean proving or disproving a theory or hypothesis, even if your research aim was to, but having done enough work to provide a useful and original insight into the principles that underlie your research aim.
2. Making Your Research Objectives Too Ambitious
Be realistic about what you can achieve in the time you have available. It is natural to want to set ambitious research objectives that require sophisticated data collection and analysis, but only completing this with six months before the end of your PhD registration period is not a worthwhile trade-off.
3. Formulating Repetitive Research Objectives
Each research objective should have its own purpose and distinct measurable outcome. To this effect, a common mistake is to form research objectives which have large amounts of overlap. This makes it difficult to determine when an objective is truly complete, and also presents challenges in estimating the duration of objectives when creating your project timeline. It also makes it difficult to structure your thesis into unique chapters, making it more challenging for you to write and for your audience to read.
Fortunately, this oversight can be easily avoided by using SMART objectives.
Hopefully, you now have a good idea of how to create an effective set of aims and objectives for your research project, whether it be a thesis, dissertation or research paper. While it may be tempting to dive directly into your research, spending time on getting your aims and objectives right will give your research clear direction. This won’t only reduce the likelihood of problems arising later down the line, but will also lead to a more thorough and coherent research project.
Finding a PhD has never been this easy – search for a PhD by keyword, location or academic area of interest.
Browse PhDs Now
Join thousands of students.
Join thousands of other students and stay up to date with the latest PhD programmes, funding opportunities and advice.
Have a language expert improve your writing
Run a free plagiarism check in 10 minutes, generate accurate citations for free.
- Knowledge Base
- Starting the research process
- Writing Strong Research Questions | Criteria & Examples
Writing Strong Research Questions | Criteria & Examples
Published on October 26, 2022 by Shona McCombes . Revised on November 21, 2023.
A research question pinpoints exactly what you want to find out in your work. A good research question is essential to guide your research paper , dissertation , or thesis .
All research questions should be:
- Focused on a single problem or issue
- Researchable using primary and/or secondary sources
- Feasible to answer within the timeframe and practical constraints
- Specific enough to answer thoroughly
- Complex enough to develop the answer over the space of a paper or thesis
- Relevant to your field of study and/or society more broadly
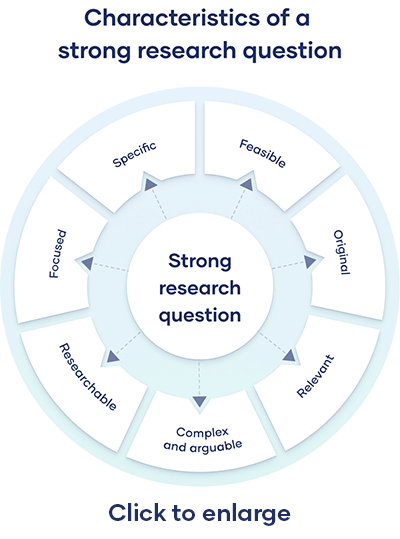
Table of contents
How to write a research question, what makes a strong research question, using sub-questions to strengthen your main research question, research questions quiz, other interesting articles, frequently asked questions about research questions.
You can follow these steps to develop a strong research question:
- Choose your topic
- Do some preliminary reading about the current state of the field
- Narrow your focus to a specific niche
- Identify the research problem that you will address
The way you frame your question depends on what your research aims to achieve. The table below shows some examples of how you might formulate questions for different purposes.
| Research question formulations | |
|---|---|
| Describing and exploring | |
| Explaining and testing | |
| Evaluating and acting | is X |
Using your research problem to develop your research question
| Example research problem | Example research question(s) |
|---|---|
| Teachers at the school do not have the skills to recognize or properly guide gifted children in the classroom. | What practical techniques can teachers use to better identify and guide gifted children? |
| Young people increasingly engage in the “gig economy,” rather than traditional full-time employment. However, it is unclear why they choose to do so. | What are the main factors influencing young people’s decisions to engage in the gig economy? |
Note that while most research questions can be answered with various types of research , the way you frame your question should help determine your choices.
Prevent plagiarism. Run a free check.
Research questions anchor your whole project, so it’s important to spend some time refining them. The criteria below can help you evaluate the strength of your research question.
Focused and researchable
| Criteria | Explanation |
|---|---|
| Focused on a single topic | Your central research question should work together with your research problem to keep your work focused. If you have multiple questions, they should all clearly tie back to your central aim. |
| Answerable using | Your question must be answerable using and/or , or by reading scholarly sources on the to develop your argument. If such data is impossible to access, you likely need to rethink your question. |
| Not based on value judgements | Avoid subjective words like , , and . These do not give clear criteria for answering the question. |
Feasible and specific
| Criteria | Explanation |
|---|---|
| Answerable within practical constraints | Make sure you have enough time and resources to do all research required to answer your question. If it seems you will not be able to gain access to the data you need, consider narrowing down your question to be more specific. |
| Uses specific, well-defined concepts | All the terms you use in the research question should have clear meanings. Avoid vague language, jargon, and too-broad ideas. |
| Does not demand a conclusive solution, policy, or course of action | Research is about informing, not instructing. Even if your project is focused on a practical problem, it should aim to improve understanding rather than demand a ready-made solution. If ready-made solutions are necessary, consider conducting instead. Action research is a research method that aims to simultaneously investigate an issue as it is solved. In other words, as its name suggests, action research conducts research and takes action at the same time. |
Complex and arguable
| Criteria | Explanation |
|---|---|
| Cannot be answered with or | Closed-ended, / questions are too simple to work as good research questions—they don’t provide enough for robust investigation and discussion. |
| Cannot be answered with easily-found facts | If you can answer the question through a single Google search, book, or article, it is probably not complex enough. A good research question requires original data, synthesis of multiple sources, and original interpretation and argumentation prior to providing an answer. |
Relevant and original
| Criteria | Explanation |
|---|---|
| Addresses a relevant problem | Your research question should be developed based on initial reading around your . It should focus on addressing a problem or gap in the existing knowledge in your field or discipline. |
| Contributes to a timely social or academic debate | The question should aim to contribute to an existing and current debate in your field or in society at large. It should produce knowledge that future researchers or practitioners can later build on. |
| Has not already been answered | You don’t have to ask something that nobody has ever thought of before, but your question should have some aspect of originality. For example, you can focus on a specific location, or explore a new angle. |
Chances are that your main research question likely can’t be answered all at once. That’s why sub-questions are important: they allow you to answer your main question in a step-by-step manner.
Good sub-questions should be:
- Less complex than the main question
- Focused only on 1 type of research
- Presented in a logical order
Here are a few examples of descriptive and framing questions:
- Descriptive: According to current government arguments, how should a European bank tax be implemented?
- Descriptive: Which countries have a bank tax/levy on financial transactions?
- Framing: How should a bank tax/levy on financial transactions look at a European level?
Keep in mind that sub-questions are by no means mandatory. They should only be asked if you need the findings to answer your main question. If your main question is simple enough to stand on its own, it’s okay to skip the sub-question part. As a rule of thumb, the more complex your subject, the more sub-questions you’ll need.
Try to limit yourself to 4 or 5 sub-questions, maximum. If you feel you need more than this, it may be indication that your main research question is not sufficiently specific. In this case, it’s is better to revisit your problem statement and try to tighten your main question up.
Here's why students love Scribbr's proofreading services
Discover proofreading & editing
If you want to know more about the research process , methodology , research bias , or statistics , make sure to check out some of our other articles with explanations and examples.
Methodology
- Sampling methods
- Simple random sampling
- Stratified sampling
- Cluster sampling
- Likert scales
- Reproducibility
Statistics
- Null hypothesis
- Statistical power
- Probability distribution
- Effect size
- Poisson distribution
Research bias
- Optimism bias
- Cognitive bias
- Implicit bias
- Hawthorne effect
- Anchoring bias
- Explicit bias
The way you present your research problem in your introduction varies depending on the nature of your research paper . A research paper that presents a sustained argument will usually encapsulate this argument in a thesis statement .
A research paper designed to present the results of empirical research tends to present a research question that it seeks to answer. It may also include a hypothesis —a prediction that will be confirmed or disproved by your research.
As you cannot possibly read every source related to your topic, it’s important to evaluate sources to assess their relevance. Use preliminary evaluation to determine whether a source is worth examining in more depth.
This involves:
- Reading abstracts , prefaces, introductions , and conclusions
- Looking at the table of contents to determine the scope of the work
- Consulting the index for key terms or the names of important scholars
A research hypothesis is your proposed answer to your research question. The research hypothesis usually includes an explanation (“ x affects y because …”).
A statistical hypothesis, on the other hand, is a mathematical statement about a population parameter. Statistical hypotheses always come in pairs: the null and alternative hypotheses . In a well-designed study , the statistical hypotheses correspond logically to the research hypothesis.
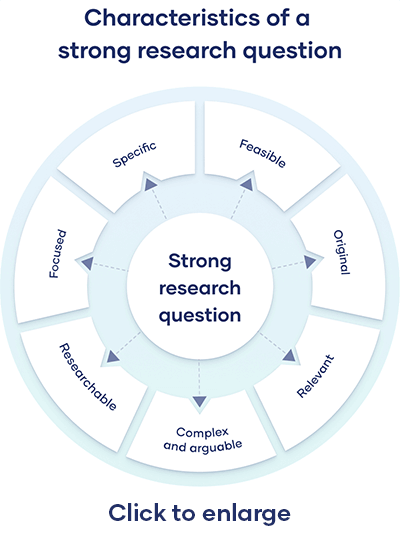
Formulating a main research question can be a difficult task. Overall, your question should contribute to solving the problem that you have defined in your problem statement .
However, it should also fulfill criteria in three main areas:
- Researchability
- Feasibility and specificity
- Relevance and originality
Cite this Scribbr article
If you want to cite this source, you can copy and paste the citation or click the “Cite this Scribbr article” button to automatically add the citation to our free Citation Generator.
McCombes, S. (2023, November 21). Writing Strong Research Questions | Criteria & Examples. Scribbr. Retrieved September 3, 2024, from https://www.scribbr.com/research-process/research-questions/
Is this article helpful?
Shona McCombes
Other students also liked, how to define a research problem | ideas & examples, how to write a problem statement | guide & examples, 10 research question examples to guide your research project, get unlimited documents corrected.
✔ Free APA citation check included ✔ Unlimited document corrections ✔ Specialized in correcting academic texts
- +23054999070
- [email protected]

Research Questions, Hypotheses and Objectives
Research questions.
A research question naturally emerges from a research problem that needs to be resolved. Developing a good research question not only rests on the study of some uncertain phenomenon, but also on the rational need for investigating it. It is therefore essential that a systematic review of extant literature on the research topic be conducted, prior to formulating a research question. Awareness of current trends and latest development in the field of study will certainly assist in formulating a relevant question. There may be several research questions, whether primary or secondary, but they should all be developed during the planning stage of a study. Furthermore, it must be ensured that secondary questions do not compromise the primary research question, which forms the basis of research objectives and hypotheses. Lastly, bear in mind that the number of research questions will inevitably make the research design and data analysis more complex.
Hulley et al . (2001) suggested the use of the FINER criteria in developing a good research question:
- F - Feasible : adequate number of subjects and technical expertise, affordability in terms of time and money, manageability in scope
- I - Interesting : getting an answer that intrigues the researcher, the peers and the community
- N - Novel : confirms, refutes or extends previous findings
- E - Ethical : a study that will be approved by an institutional review board
- R - Relevant : to scientific knowledge and future research
A poorly formulated research question may thus affect the choice of study design and hamper the chance of obtaining any significant finding, besides compromising the quality of the study.
Research Hypotheses
First of all, it is essential to understand that it is a hypothesis, not the data, that drives a primary research question . Otherwise, given any dataset, it would be too simple to perform several tests and apply statistical techniques to establish significant associations and/or relationships among variables and/or constructs. In such cases, it would be working backward by using the data to develop the research question, and that would defeat the entire purpose of conducting the study. To make matters worse, spuriously positive findings may result.
Hypothesis testing aims at making inferences about the targeted population on the basis of a random sample taken from that population. A hypothesis must be formulated as a null hypothesis, generally meaning that a prevailing situation has not changed (in the case of finding differences) or that there are no significant relationships among variables and/or constructs. This is the reason why each null hypoythesis must be paired with an alternative hypothesis, should the outcome be significant. The two hypotheses must be mutually exclusive and comprehensively exhaustive, i.e., the acceptance of one would automatically imply the rejection of the other. For a better understanding of the concept of hypothesis testing, you might need to consult our statistician.
At this stage, all you need to know is that the development of a research hypothesis should be supported by a good research question, as it will influence your research design. Once appropriate hypotheses have been developed, you can safely proceed to the formulation of your research objectives.
Research Objectives
You must first learn to distinguish between a research aim and a research objective . While an aim is written in broad terms and explains what is to be achieved at the end of the study, an objective is an active statement that is defined in measurable outcomes via a strong positive statement. The primary objective of a study is paired with the hypothesis of the study, and should be clearly stated in the introduction of the research protocol. Objectives usually state exactly the outcome measures that are going to be used within their statements. Strong verbs like determine , measure , assess , evaluate , identify , examine , investigate , etc., are used in the formulation of objectives.
The importance of objectives is that they guide the development of the protocol and design of study, and play a determining role in sample size calculations. Objectives should be focused on outcomes that are important and relevant to the study.
Research aim To investigate the issue of student indiscipline and its impact on student attainment in Mauritian Secondary Schools Research question What are the various types of student indiscipline currently experienced in secondary schools of Mauritius? Research (null) hypothesis School management style does not impact on student attainment Research objective To identify the most common forms of indiscipline and their level of seriousness

Copyright © 2024, Stat Consul, All rights reserved. Developed by Algopage

How to Develop a Good Research Hypothesis
The story of a research study begins by asking a question. Researchers all around the globe are asking curious questions and formulating research hypothesis. However, whether the research study provides an effective conclusion depends on how well one develops a good research hypothesis. Research hypothesis examples could help researchers get an idea as to how to write a good research hypothesis.
This blog will help you understand what is a research hypothesis, its characteristics and, how to formulate a research hypothesis
Table of Contents
What is Hypothesis?
Hypothesis is an assumption or an idea proposed for the sake of argument so that it can be tested. It is a precise, testable statement of what the researchers predict will be outcome of the study. Hypothesis usually involves proposing a relationship between two variables: the independent variable (what the researchers change) and the dependent variable (what the research measures).
What is a Research Hypothesis?
Research hypothesis is a statement that introduces a research question and proposes an expected result. It is an integral part of the scientific method that forms the basis of scientific experiments. Therefore, you need to be careful and thorough when building your research hypothesis. A minor flaw in the construction of your hypothesis could have an adverse effect on your experiment. In research, there is a convention that the hypothesis is written in two forms, the null hypothesis, and the alternative hypothesis (called the experimental hypothesis when the method of investigation is an experiment).
Characteristics of a Good Research Hypothesis
As the hypothesis is specific, there is a testable prediction about what you expect to happen in a study. You may consider drawing hypothesis from previously published research based on the theory.
A good research hypothesis involves more effort than just a guess. In particular, your hypothesis may begin with a question that could be further explored through background research.
To help you formulate a promising research hypothesis, you should ask yourself the following questions:
- Is the language clear and focused?
- What is the relationship between your hypothesis and your research topic?
- Is your hypothesis testable? If yes, then how?
- What are the possible explanations that you might want to explore?
- Does your hypothesis include both an independent and dependent variable?
- Can you manipulate your variables without hampering the ethical standards?
- Does your research predict the relationship and outcome?
- Is your research simple and concise (avoids wordiness)?
- Is it clear with no ambiguity or assumptions about the readers’ knowledge
- Is your research observable and testable results?
- Is it relevant and specific to the research question or problem?

The questions listed above can be used as a checklist to make sure your hypothesis is based on a solid foundation. Furthermore, it can help you identify weaknesses in your hypothesis and revise it if necessary.
Source: Educational Hub
How to formulate a research hypothesis.
A testable hypothesis is not a simple statement. It is rather an intricate statement that needs to offer a clear introduction to a scientific experiment, its intentions, and the possible outcomes. However, there are some important things to consider when building a compelling hypothesis.
1. State the problem that you are trying to solve.
Make sure that the hypothesis clearly defines the topic and the focus of the experiment.
2. Try to write the hypothesis as an if-then statement.
Follow this template: If a specific action is taken, then a certain outcome is expected.
3. Define the variables
Independent variables are the ones that are manipulated, controlled, or changed. Independent variables are isolated from other factors of the study.
Dependent variables , as the name suggests are dependent on other factors of the study. They are influenced by the change in independent variable.
4. Scrutinize the hypothesis
Evaluate assumptions, predictions, and evidence rigorously to refine your understanding.
Types of Research Hypothesis
The types of research hypothesis are stated below:
1. Simple Hypothesis
It predicts the relationship between a single dependent variable and a single independent variable.
2. Complex Hypothesis
It predicts the relationship between two or more independent and dependent variables.
3. Directional Hypothesis
It specifies the expected direction to be followed to determine the relationship between variables and is derived from theory. Furthermore, it implies the researcher’s intellectual commitment to a particular outcome.
4. Non-directional Hypothesis
It does not predict the exact direction or nature of the relationship between the two variables. The non-directional hypothesis is used when there is no theory involved or when findings contradict previous research.
5. Associative and Causal Hypothesis
The associative hypothesis defines interdependency between variables. A change in one variable results in the change of the other variable. On the other hand, the causal hypothesis proposes an effect on the dependent due to manipulation of the independent variable.
6. Null Hypothesis
Null hypothesis states a negative statement to support the researcher’s findings that there is no relationship between two variables. There will be no changes in the dependent variable due the manipulation of the independent variable. Furthermore, it states results are due to chance and are not significant in terms of supporting the idea being investigated.
7. Alternative Hypothesis
It states that there is a relationship between the two variables of the study and that the results are significant to the research topic. An experimental hypothesis predicts what changes will take place in the dependent variable when the independent variable is manipulated. Also, it states that the results are not due to chance and that they are significant in terms of supporting the theory being investigated.
Research Hypothesis Examples of Independent and Dependent Variables
Research Hypothesis Example 1 The greater number of coal plants in a region (independent variable) increases water pollution (dependent variable). If you change the independent variable (building more coal factories), it will change the dependent variable (amount of water pollution).
Research Hypothesis Example 2 What is the effect of diet or regular soda (independent variable) on blood sugar levels (dependent variable)? If you change the independent variable (the type of soda you consume), it will change the dependent variable (blood sugar levels)
You should not ignore the importance of the above steps. The validity of your experiment and its results rely on a robust testable hypothesis. Developing a strong testable hypothesis has few advantages, it compels us to think intensely and specifically about the outcomes of a study. Consequently, it enables us to understand the implication of the question and the different variables involved in the study. Furthermore, it helps us to make precise predictions based on prior research. Hence, forming a hypothesis would be of great value to the research. Here are some good examples of testable hypotheses.
More importantly, you need to build a robust testable research hypothesis for your scientific experiments. A testable hypothesis is a hypothesis that can be proved or disproved as a result of experimentation.
Importance of a Testable Hypothesis
To devise and perform an experiment using scientific method, you need to make sure that your hypothesis is testable. To be considered testable, some essential criteria must be met:
- There must be a possibility to prove that the hypothesis is true.
- There must be a possibility to prove that the hypothesis is false.
- The results of the hypothesis must be reproducible.
Without these criteria, the hypothesis and the results will be vague. As a result, the experiment will not prove or disprove anything significant.
What are your experiences with building hypotheses for scientific experiments? What challenges did you face? How did you overcome these challenges? Please share your thoughts with us in the comments section.
Frequently Asked Questions
The steps to write a research hypothesis are: 1. Stating the problem: Ensure that the hypothesis defines the research problem 2. Writing a hypothesis as an 'if-then' statement: Include the action and the expected outcome of your study by following a ‘if-then’ structure. 3. Defining the variables: Define the variables as Dependent or Independent based on their dependency to other factors. 4. Scrutinizing the hypothesis: Identify the type of your hypothesis
Hypothesis testing is a statistical tool which is used to make inferences about a population data to draw conclusions for a particular hypothesis.
Hypothesis in statistics is a formal statement about the nature of a population within a structured framework of a statistical model. It is used to test an existing hypothesis by studying a population.
Research hypothesis is a statement that introduces a research question and proposes an expected result. It forms the basis of scientific experiments.
The different types of hypothesis in research are: • Null hypothesis: Null hypothesis is a negative statement to support the researcher’s findings that there is no relationship between two variables. • Alternate hypothesis: Alternate hypothesis predicts the relationship between the two variables of the study. • Directional hypothesis: Directional hypothesis specifies the expected direction to be followed to determine the relationship between variables. • Non-directional hypothesis: Non-directional hypothesis does not predict the exact direction or nature of the relationship between the two variables. • Simple hypothesis: Simple hypothesis predicts the relationship between a single dependent variable and a single independent variable. • Complex hypothesis: Complex hypothesis predicts the relationship between two or more independent and dependent variables. • Associative and casual hypothesis: Associative and casual hypothesis predicts the relationship between two or more independent and dependent variables. • Empirical hypothesis: Empirical hypothesis can be tested via experiments and observation. • Statistical hypothesis: A statistical hypothesis utilizes statistical models to draw conclusions about broader populations.
Wow! You really simplified your explanation that even dummies would find it easy to comprehend. Thank you so much.
Thanks a lot for your valuable guidance.
I enjoy reading the post. Hypotheses are actually an intrinsic part in a study. It bridges the research question and the methodology of the study.
Useful piece!
This is awesome.Wow.
It very interesting to read the topic, can you guide me any specific example of hypothesis process establish throw the Demand and supply of the specific product in market
Nicely explained
It is really a useful for me Kindly give some examples of hypothesis
It was a well explained content ,can you please give me an example with the null and alternative hypothesis illustrated
clear and concise. thanks.
So Good so Amazing
Good to learn
Thanks a lot for explaining to my level of understanding
Explained well and in simple terms. Quick read! Thank you
It awesome. It has really positioned me in my research project
Brief and easily digested
Rate this article Cancel Reply
Your email address will not be published.

Enago Academy's Most Popular Articles

- Reporting Research
Choosing the Right Analytical Approach: Thematic analysis vs. content analysis for data interpretation
In research, choosing the right approach to understand data is crucial for deriving meaningful insights.…

Comparing Cross Sectional and Longitudinal Studies: 5 steps for choosing the right approach
The process of choosing the right research design can put ourselves at the crossroads of…

- Industry News
COPE Forum Discussion Highlights Challenges and Urges Clarity in Institutional Authorship Standards
The COPE forum discussion held in December 2023 initiated with a fundamental question — is…

- Career Corner
Unlocking the Power of Networking in Academic Conferences
Embarking on your first academic conference experience? Fear not, we got you covered! Academic conferences…

Research Recommendations – Guiding policy-makers for evidence-based decision making
Research recommendations play a crucial role in guiding scholars and researchers toward fruitful avenues of…
Choosing the Right Analytical Approach: Thematic analysis vs. content analysis for…
Comparing Cross Sectional and Longitudinal Studies: 5 steps for choosing the right…
How to Design Effective Research Questionnaires for Robust Findings

Sign-up to read more
Subscribe for free to get unrestricted access to all our resources on research writing and academic publishing including:
- 2000+ blog articles
- 50+ Webinars
- 10+ Expert podcasts
- 50+ Infographics
- 10+ Checklists
- Research Guides
We hate spam too. We promise to protect your privacy and never spam you.
- Publishing Research
- AI in Academia
- Promoting Research
- Diversity and Inclusion
- Infographics
- Expert Video Library
- Other Resources
- Enago Learn
- Upcoming & On-Demand Webinars
- Peer Review Week 2024
- Open Access Week 2023
- Conference Videos
- Enago Report
- Journal Finder
- Enago Plagiarism & AI Grammar Check
- Editing Services
- Publication Support Services
- Research Impact
- Translation Services
- Publication solutions
- AI-Based Solutions
- Thought Leadership
- Call for Articles
- Call for Speakers
- Author Training
- Edit Profile
I am looking for Editing/ Proofreading services for my manuscript Tentative date of next journal submission:

In your opinion, what is the most effective way to improve integrity in the peer review process?
Research questions and research objectives.
- January 2005

- University of Malaya
Discover the world's research
- 25+ million members
- 160+ million publication pages
- 2.3+ billion citations

- Maurice Ndjouma Wedjou

- Alain Vilard
- Femi MIchael NEJO

- Keng Yin Loh

- Prim Health Care Res Dev
- John W Beasley

- Jan P Vandenbroucke

- New Zeal Fam Physician
- M Kljakovic
- W B Michael
- Recruit researchers
- Join for free
- Login Email Tip: Most researchers use their institutional email address as their ResearchGate login Password Forgot password? Keep me logged in Log in or Continue with Google Welcome back! Please log in. Email · Hint Tip: Most researchers use their institutional email address as their ResearchGate login Password Forgot password? Keep me logged in Log in or Continue with Google No account? Sign up
Part 1. Overview Information
National Institutes of Health ( NIH )
National Institute on Aging ( NIA )
National Institute on Alcohol Abuse and Alcoholism ( NIAAA )
National Institute of Arthritis and Musculoskeletal and Skin Diseases ( NIAMS )
Eunice Kennedy Shriver National Institute of Child Health and Human Development ( NICHD )
National Institute of Neurological Disorders and Stroke ( NINDS )
R15 Research Enhancement Award (REA)
- April 4, 2024 - Overview of Grant Application and Review Changes for Due Dates on or after January 25, 2025. See Notice NOT-OD-24-084 .
- August 31, 2022 - Implementation Changes for Genomic Data Sharing Plans Included with Applications Due on or after January 25, 2023. See Notice NOT-OD-22-198 .
- August 5, 2022 - Implementation Details for the NIH Data Management and Sharing Policy. See Notice NOT-OD-22-189 .
See Part 2, Section III. 3. Additional Information on Eligibility.
The purpose of this HEAL Initiative program is to: (1) support basic and mechanistic pain research from R15-eligible undergraduate-focused serving institutions, health professional schools or graduate schools; (2) promote integrated, interdisciplinary research partnerships between Principal Investigators (PIs) from R15-eligible institutions and investigators from U.S. domestic institutions; and (3) enhance the pain research environment at the R15-eligible institution for health professional students, undergraduate and/or graduate students through active engagement in pain research.
Applications in response to this notice of funding opportunity (NOFO) should include plans to accomplish these goals. Specifically, applications should include a rigorous plan for conducting basic and mechanistic pain research in the Research Strategy section of the application . In addition, a research partnership between the PI’s institution and at least one investigator from a separate U.S. domestic institution that provides resources and/or expertise that will enhance the proposed pain research program must be included in a separate Team Management Plan. The proposed partnership will be a sub-award agreement(s) with at least one partnering institution, which does not need to be R15-eligible. The budget of all sub-awards must not exceed one third of the total budget. Furthermore, applications must include a Facilities & Other Resources document that demonstrates active involvement of health professional students, undergraduate and/or graduate students from the R15-eligible institution(s) in the proposed pain research projects.
This Notice of Funding Opportunity (NOFO) requires a Plan for Enhancing Diverse Perspectives (PEDP).
30 days before application due date.
| Application Due Dates | Review and Award Cycles | ||||
|---|---|---|---|---|---|
| New | Renewal / Resubmission / Revision (as allowed) | AIDS - New/Renewal/Resubmission/Revision, as allowed | Scientific Merit Review | Advisory Council Review | Earliest Start Date |
| November 19, 2024 | Not Applicable | December 18, 2024 | March 2025 | May 2025 | July 2025 |
| October 28, 2025 | October 28, 2025 | November 24, 2025 | March 2026 | May 2026 | July 2026 |
| October 27, 2026 | October 27, 2026 | November 23, 2026 | March 2027 | May 2027 | July 2027 |
All applications are due by 5:00 PM local time of applicant organization.
Applicants are encouraged to apply early to allow adequate time to make any corrections to errors found in the application during the submission process by the due date.
No late applications will be accepted for this Notice of Funding Opportunity (NOFO).
Not Applicable
It is critical that applicants follow the instructions in the Research (R) Instructions in the How to Apply - Application Guide , except where instructed to do otherwise (in this NOFO or in a Notice from NIH Guide for Grants and Contracts ).
Conformance to all requirements (both in the How to Apply - Application Guide and the NOFO) is required and strictly enforced. Applicants must read and follow all application instructions in the How to Apply - Application Guide as well as any program-specific instructions noted in Section IV. When the program-specific instructions deviate from those in the How to Apply - Application Guide , follow the program-specific instructions.
Applications that do not comply with these instructions may be delayed or not accepted for review.
There are several options available to submit your application through Grants.gov to NIH and Department of Health and Human Services partners. You must use one of these submission options to access the application forms for this opportunity.
- Use the NIH ASSIST system to prepare, submit and track your application online.
- Use an institutional system-to-system (S2S) solution to prepare and submit your application to Grants.gov and eRA Commons to track your application. Check with your institutional officials regarding availability.
- Use Grants.gov Workspace to prepare and submit your application and eRA Commons to track your application.
Part 2. Full Text of Announcement
Section i. notice of funding opportunity description.
Applications in response to this notice of funding opportunity (NOFO) should include plans to accomplish these goals. Specifically, applications should include a rigorous plan for conducting basic and mechanistic pain research projects in the Research Strategy section of the application. In addition, a research partnership between the PI’s institution and at least one investigator from a separate U.S. domestic institution that provides resources and/or expertise that will enhance the proposed pain research program must be included in a separate Team Management Plan. The proposed partnership will be a sub-award agreement(s) with at least one partnering institution, which does not need to be R15-eligible. The budget of all sub-awards must not exceed one third of the total budget. Furthermore, applications must include a Facilities & Other Resources document that demonstrates active involvement of health professional students or undergraduate and/or graduate students from the R15-eligible institution(s) in the proposed pain research projects.
The National Institutes of Health (NIH) Helping to End Addiction Long-term ® Initiative, or NIH HEAL Initiative ® , bolsters research across NIH to (1) improve treatment for opioid misuse and addiction and (2) enhance pain management. More information about the NIH HEAL Initiative is available at https://heal.nih.gov/ . Research shows that diverse teams working together and capitalizing on innovative ideas and distinct perspectives outperform homogeneous teams. Scientists and trainees from diverse backgrounds and life experiences bring different perspectives, creativity, and individual enterprise to address complex scientific problems. See the Notice of NIH’s Interest in Diversity ( NOT-OD-20-031 ) for more details. Promoting diversity in the pain research workforce is crucial to promoting future scientific advances in this area and to achieve the NIH HEAL Initiative’s workforce development goals.The initiative has funded multiple pain workforce enhancement programs that support early-career investigators. Despite these efforts, the NIH HEAL Initiative can benefit from additionally supporting R-15 eligible institutions that involve undergraduate, graduate or health professional school/colleges students in pain research.
Since Fiscal Year (FY) 1985, NIH has made a special effort to stimulate research at educational institutions that provide baccalaureate and/or advanced degrees for a significant number of the nation’s research scientists who have not been major recipients of NIH support. NIH has implemented two parent award programs, the Academic Research Enhancement Award (AREA) program ( PAR-21-155 ) and Research Enhancement Award Program (REAP) ( PAR-22-060 ), to provide research experiences to health professional or undergraduate and/or graduate students pursuing biomedical or behavioral research at U.S. higher education institutions. Utilizing these two programs will further promote a diverse pain research workforce. This Pain Research Enhancement Program (“PREP”) will further support meritorious collaborative pain research from designated educational levels in the NIH HEAL Initiative, using the NIH Research Enhancement Award programs as a guide. Specifically, this NOFO aims to support new scientific solutions to the national opioid public health crisis byestablishing new research partnerships that will lead to research experiences for undergraduate, graduate, and health professional students, to further enhance the pool of potential participants in the pain research pipeline.
Program Objectives:
The purpose of this HEAL Initiative program is to: (1) support basic and mechanistic pain research from R15-eligible undergraduate-focused serving institutions, health professional schools or graduate schools; (2) promote integrated, interdisciplinary research partnerships between Principal Investigators (PIs) from R15-eligible institutions and investigators from U.S. domestic institutions; and (3) enhance the pain research environment at the R15-eligible institution for health professional students, undergraduate and/or graduate students through active engagement in pain research. Successful applications will include plans detailing how they intend to accomplish all three goals. Please refer to Section III for specific R15 eligibility information. Although preliminary data are not required for an R15 application, they may be included if available. The scientific foundation for the proposed research should be based on published research and/or any available preliminary data.
Objective 1: Develop Small-Scale Basic and Mechanistic Pain Research Projects
Proposed research projects should be hypothesis driven and use a rigorous scientific design to generate research data/evidence and advance scientific knowledge. Applications should include objectives that are attainable within the 3-year grant period.
Pain research projects may include, but are not limited to, the study of: nociception and/or pain processing in non-pain populations, acute pain, cancer pain, chemotherapy-induced neuropathy, chronic pain, diabetic neuropathy, eye pain, gynecologic pain, headache, musculoskeletal pain, myofascial pain, obstetric pain, osteoarthritis, pain conditions across the lifespan (including in the context of aging), pain co-occurring with substance use disorders (SUDs), painful disorders of the orofacial region, painful neuropathy, post-stroke pain, post-surgical pain, sickle cell pain, and/or visceral pain. Innovative pain research topics that propose an interdisciplinary mechanistic pain research are considered high program priority under this initiative.
Projects may focus on basic pain research with pre-clinical ( e.g., animal or in silico ) models or involve research participants ( e.g., observational studies, epidemiological studies, secondary data analyses, or device development). Alternatively, investigators may propose a mechanistic and/or “Basic Experimental Studies involving Humans” (BESH) clinical trial as described below. Clinical trials designed primarily to determine the safety, tolerability, and/or clinical efficacy of an intervention will be considered non-responsive to this NOFO and withdrawn without review .
For this NOFO, only the following types of clinical trials will be supported:
- Basic Experimental Studies with Humans (BESH) , defined as basic research studies involving humans that seek to understand the fundamental aspects of phenomena
- Mechanistic trials , defined as studies designed to understand a biological or behavioral process, the pathophysiology of a disease, or the mechanism of action of an intervention (i.e., how an intervention works, but not if it works or is safe)
NIH defines a clinical trial as a research study in which one or more human subjects are prospectively assigned to one or more interventions (which may include placebo or other control) to evaluate the effects of those interventions on health-related biomedical or behavioral outcomes ( https://grants.nih.gov/grants/guide/notice-files/NOT-OD-15-015.html ). For further clarification on how NIH defines the different types of clinical trials, please refer to the following resources:
- NOT-OD-15-015: Notice of Revised NIH Definition of Clinical Trial
- NIH's Definition of a Clinical Trial
- Decision Tree for NIH Clinical Trial Definition
- Guidance for Basic Experimental Studies with Humans (BESH) Funding Opportunities
- NIH Definition of Clinical Trial Case Studies
Objective 2: Promote Integrated, Interdisciplinary Research Partnerships
A second key objective of this NOFO is to promote new research partnerships among investigators at R15-eligible institutions with separate (legally distinct) investigators at domestic research institutions. Investigators can have a multitude of research expertise that aligns with the proposed research projects and/or resources that can be shared to enhance the proposed research. Applications must propose a collaboration with at least one sub-award holder from a separate U.S. domestic research institution and should include details of how the collaboration will enhance the R15-research program must be described. Applications are permitted to have a subaward to a non-R15-eligible institution. However, it is expected that PD/PI(s) from R15-eligible institution(s) will lead the proposed project and complete most of the research at the R15-eligible institution. As such, PI(s) from R15-eligible institutions must serve as the contact program director (PD)/PI for the project. Additionally, no more than one third of the total budget for the project may be used by the identified sub-award institution.
Applications that propose new interdisciplinary are considered a high program priority under this NOFO. Interdisciplinary partnership could include, but are not limited to, any two or more areas of research expertise from the following:
- Clinical pain management (e.g., nonpharmacologic or pharmacologic interventions)
- Clinical pain research
- Preclinical/basic pain biology and modeling
- Specific disease and/or pathological conditions (either human or preclinical models)
- Animal behavior
- Artificial intelligence
- Data science
In addition, a Team Management Plan is required as part of Objective 2. Studies of team science have highlighted the need for effective management structures to achieve program goals. Many resources exist to aid in developing effective team-based programs (e.g., the National Cancer Institute Collaboration and Team Science Field Guide ). The Team Management Plan focuses on management of the whole team/key personnel. Because teams will likely include individuals from widely divergent scientific backgrounds, teams must have a shared vision and a defined plan for communication and management of shared responsibilities, interpersonal interactions, and professional credit. The Team Management Plan should be included as an attachment (three pages maximum) to this application. It should address how the research team, including the PI from R15-eligible Instiution and collaborator(s), will work together to accomplish program objectives. See the application instructions for “Other Attachments” on the SF424(R&R) Other Project Information in Section IV.2 Instructions for Application Submission for details. The Team Management Plan should address the following points:
- Organizational structure and team composition and roles
- Shared leadership, contributions, and distributed responsibility for decision-making
- Resource sharing and allocation
- Credit assignment and/or intellectual property (IP) rights
- Coordination and communication plans
- Intra-team data sharing, archiving, and preservation
Objective 3: Enhance the Research Environment by Engaging Students
The third objective of this program is to enhance the pain research environment at the R15-eligible institution by engaging and providing research opportunities to health professional students or undergraduate and/or graduate students. A Facilities & Other Resources document is required to describe how the proposed research will enhance the pain research environment at the R15-eligible institution. Two-thirds of the proposed research project team should comprise personnel from the R15-eligible institutions, including health professional students, or graduate students or undergraduate students from the primary R15-eligible institution. Although the proposed research project must be led by the identified PD/PI, applications with strong and innovative student engagement are of high program priority. If participating students have not yet been identified, the number and academic stage of those to be involved should be provided. Applications should identify which aspects of the proposed research will include student participation. Student involvement may include participation in the design of experiments, collection and analysis of data, execution and troubleshooting of experiments, participation in research meetings, and discussion of future research directions. When applicable, it is highly desirable that student participation also include presentation of research at local and/or national meetings (including the HEAL Annual Scientific Meeting and "Positively Uniting Researchers of Pain to Opine, Synthesize, & Engage" {PURPOSE} meeting), publication of journal articles, and collaborative interactions. By engaging in these activities and collaborating on pain-focused research projects at early stages of training, students will be better prepared and motivated to pursue careers in pain research. Please see Section III for a list of eligible students.
This NOFO aims to support pain research grants, not training or fellowship program s. As such, applications should not include training plans such as didactic training or non-research activities related to professional development. Likewise, applications should not include independent student research projects. For applications that propose a clinical trial, the PD/PI must be the responsible individual of record for oversight of the trial though students can take part in all components of a clinical trial. Oversight includes (but is not limited to): interacting with relevant Institutional Review Board (IRB) staff; reviewing all informed consent documents; reporting potential serious adverse events; and maintaining responsibility for patient safety. However, the student can gain experience in all these components in conjunction with the individual leading the trial. Applications submitted to this NOFO may include additional investigators to those outlined above, including additional collaborators or consultants, or other individuals such as high school students, post-baccalaureate participants, postdoctoral fellows, or clinical fellows. However, involvement of such individuals does not fulfill the goal of enhancing the R15-eligible institutional environment and should account for less than one third of the overall proposed project team.
Additional Information
Non-responsiveness Criteria:
Applications deemed to be non-responsive will not proceed to review and will be withdrawn. Applications with one or more of the following characteristics are considered non-responsive to this NOFO:
- Research that does not address the NIH HEAL Initiative mission to enhance pain management.
- Failure to describe a proposed Research plan and specific aims primarily led by a PI from a R15-eligible Institution.
- Omission of a domestic research partnership and accompanying sub award(s), or that include sub-award(s) that account for more than one third of the total project budget.
- Failure to include the required Facilities & Other Resources document and Other Attachments, including a Team Management Planand, letters of support, including a letter of support from the identified subaward holder(s) and a letter of support from the R15-eligible institution’s provost. Please see s ection IV.2 “Instructions for Application Submission” for details.
- Proposing a clinical trial addressing safety, tolerability, efficacy, and/or effectiveness of pharmacologic, behavioral, biologic, surgical, or device (invasive or noninvasive) interventions.
Contacting Program Officers Prior to Submission
Applicants are strongly encouraged to consult with program staff as plans for an application are being developed.
Rigor and Reproducibility
NIH strives for rigor and transparency in all research it funds. For this reason, the NIH HEAL Initiative explicitly emphasizes the NIH application instructions related to rigor and transparency ( https://grants.nih.gov/policy/reproducibility/guidance.htm ) and provides additional guidance from individual NIH institutes and centers (ICs) to the scientific community. For example, the biological rationale for the proposed experiments must be based on rigorous and robust supporting data, which means that data should be collected via methods that minimize the risk of bias and be reported in a transparent manner. If previously published or preliminary studies do not meet these standards, applicants should address how the current study design addresses the deficiencies in rigor and transparency. Proposed experiments should likewise be designed in a manner that minimizes the risk of bias and ensures validity of experimental results.
Proposed research projects should incorporate adequate methodological rigor where applicable, including but not limited to a clear rationale for the chosen model(s) and primary/secondary endpoint(s), clear descriptions of tools and parameters, blinding, randomization, adequate sample size, prespecified inclusion/exclusion criteria, appropriate handling of missing data and outliers, appropriate controls, pre-planned analyses, and appropriate quantitative techniques.
Applications should also clearly indicate the exploratory vs. confirmatory components of the study, consider study limitations, and plan for transparent reporting of all methods, analyses, and results so that other investigators can evaluate the quality of the work and potentially perform replications. NIH intends to maximize the impact of NIH HEAL Initiative-supported projects through broad and rapid data sharing and immediate access to publications ( https://heal.nih.gov/about/public-access-data ). Guidelines for complying with the HEAL Public Access and Data Sharing Policy can be found at https://heal.nih.gov/data/complying-heal-data-sharing-policy . More details about NIH HEAL Initiative data sharing are described in Section IV.
Clinical Trial Accrual Policy:
For applications that are proposing to conduct a clinical trial, a series of clinical recruitment milestones detailing completion of the clinical trial and providing contingency plans to proactively confront potential delays or disturbances in attaining the clinical recruitment milestones must be included along with a study timeline in the PHS Human Subjects and Clinical Trials Information form. Continuation of the award is conditional upon satisfactory progress, availability of funds, and scientific priorities of the NIH HEAL Initiative. If, at any time, recruitment falls significantly below the projected milestones for recruitment, NIH will consider ending support and negotiating an orderly phaseout of the award. NIH retains the option of periodic external peer review of progress. NIH program staff will closely monitor progress at all stages for milestones, accrual, and safety.
Expected Activities of Coordination
NIH HEAL Initiative awardees are strongly encouraged to cooperate and coordinate their activities. It is expected that NIH HEAL Initiative awardees will cooperate and coordinate their activities after post award by participating in PD/PI meetings, including:
NIH HEAL Initiative Scientific Meeting Attendance
Applicants and students are highly encouraged to attend the annual NIH HEAL Initiative Scientific Meetings. The NIH HEAL Initiative hosts an annual meeting of more than 800 NIH HEAL Initiative-funded researchers across the initiative’s research portfolio and career stage spectrum, NIH staff, people with lived and living experience, community partners advising initiative-funded projects, advocacy groups, and other stakeholders to
- Share research advances and cutting-edge science
- Discover opportunities, challenges, and approaches to build on the initiative’s progress
- Connect and explore collaboration with other NIH HEAL Initiative-funded researchers and collaborators to enhance initiative-funded research.
Annual National Pain Scientists Career Development Program (PURPOSE) Meeting
Applicants and students are also highly encouraged to enroll in the HEAL Initiative: Positively Uniting Researchers of Pain to Opine, Synthesize, and Engage (PURPOSE) network and attend its annual meetings. Details can be found at https://painresearchers.com . The HEAL R24 Coordinating Center for National Pain Scientists works to improve the collaboration between basic, translational, and clinical researchers who do not regularly collaborate or work together. One function of the HEAL R24 Coordinating Center for National Pain Scientists is to organize an annual meeting for established scientists as well as early-career pain investigators. This annual meeting facilitates the creation of a network of pain research mentors and mentees as well as fostering communication between scientists and clinicians of different disciplines and providing enhanced mentorship, leadership courses, and any additional training that might be helpful for early-career scientists. R15 recipients are encouraged to attend the annual PURPOSE meeting, either virtually or in person.
See Section VIII. Other Information for award authorities and regulations.
Plan for Enhancing Diverse Perspectives (PEDP) The NIH recognizes that teams comprised of investigators with diverse perspectives working together and capitalizing on innovative ideas and distinct viewpoints outperform homogeneous teams. There are many benefits that flow from a scientific workforce rich with diverse perspectives, including: fostering scientific innovation, enhancing global competitiveness, contributing to robust learning environments, improving the quality of the research, advancing the likelihood that underserved populations participate in, and benefit from research, and enhancing public trust. To support the best science, the NIH encourages inclusivity in research guided by the consideration of diverse perspectives. Broadly, diverse perspectives can include but are not limited to the educational background and scientific expertise of the people who perform the research; the populations who participate as human subjects in research studies; and the places where research is done. This NOFO requires a Plan for Enhancing Diverse Perspectives (PEDP), which will be assessed as part of the scientific and technical peer review evaluation. Assessment of applications containing a PEDP are based on the scientific and technical merit of the proposed project. Consistent with federal law, the race, ethnicity, or sex (including gender identify, sexual orientation, or transgender status) of a researcher, award participant, or trainee will not be considered during the application review process or when making funding decisions. Applications that fail to include a PEDP will be considered incomplete and will be administratively withdrawn before review. The PEDP will be submitted as Other Project Information as an attachment (see Section IV). Applicants are strongly encouraged to read the NOFO instructions carefully and view the available PEDP guidance materials .
Investigators proposing NIH-defined clinical trials may refer to the Research Methods Resources website for information about developing statistical methods and study designs.
Section II. Award Information
Grant: A financial assistance mechanism providing money, property, or both to an eligible entity to carry out an approved project or activity.
The OER Glossary and the How to Apply - Application Guide provide details on these application types. Only those application types listed here are allowed for this NOFO.
Optional: Accepting applications that either propose or do not propose clinical trial(s).
Need help determining whether you are doing a clinical trial?
The NIH HEAL Initiative intends to commit an estimated total of $1.25 million to fund up to three awards per year for FY 2025, FY 2026, and FY 2027. Support for this funding opportunity is contingent upon annual NIH appropriations and the submission of a sufficient number of meritorious applications
Applicants may request up to $375,000 in direct costs for the entire project period. No more than one third of total project costs may go to non-R15-eligible institutions. Annual inflationary increases are not allowed.
The scope of the proposed project should determine the project period. The maximum project period is 3 years.
NIH grants policies as described in the NIH Grants Policy Statement will apply to the applications submitted and awards made from this NOFO.
Section III. Eligibility Information
1. eligible applicants eligible organizations higher education institutions public/state controlled institutions of higher education private institutions of higher education the following types of higher education institutions are always encouraged to apply for nih support as public or private institutions of higher education: hispanic-serving institutions historically black colleges and universities (hbcus) tribally controlled colleges and universities (tccus) alaska native and native hawaiian serving institutions asian american native american pacific islander serving institutions (aanapisis) in addition, applicant organizations must meet the following criteria at the time of submission: the applicant organization must be an accredited public or nonprofit private school that grants baccalaureate or advanced degrees in health professions (see section below for more details) or biomedical and behavioral sciences. the application must be submitted by the eligible organization with a unique entity identifier (such as uei or duns) and a unique nih era institutional profile file (ipf) number. at the time of application submission, determination of eligibility will be based in part on nih institutional support. a year is defined as a federal fiscal year: from october 1 through september 30. note that collaborating subawardees do not need to adhere to the r15 eligibility criteria stated above. however, they must be separate legal entities that fulfill the terms of an eligible subaward agreement. for this particular nofo, they must also be u.s. domestic institutions. more details can be found at https://grants.nih.gov/policy/subawards . undergraduate focused institutions: at the time of application submission, all the non-health professional components of the institution combined must not have received support from the nih totaling more than $6 million per year (in both direct and f&a/indirect costs) in 4 of the last 7 years. for institutions composed of multiple schools and colleges, the $6 million funding limit is based on the amount of nih funding received by all the non-health professional schools and colleges within the institution as a whole. note that all activity codes are included in this calculation except the following: c06, s10, and all activity codes starting with a g. help determining the organization funding level can be found at https://grants.nih.gov/grants/funding/determing-organization-funding-levels-r15-eligibility.pdf an academic component is any school/college that is not a health professional school or college. a qualifying academic component (i.e., school/college) within an institution (e.g., school of arts and sciences) has greater undergraduate student enrollment than graduate student enrollment. all types of health professional schools and colleges are not eligible to apply and are not considered in this calculation. for institutions with multiple campuses, eligibility can be considered for each individual campus (e.g., main, satellite, etc.) only if separate ueis and nih ipf numbers are established for each campus. for institutions that use one uei or nih ipf number for all campuses, eligibility is determined for all campuses (e.g., main, satellite, etc.) combined. health professional and graduate schools at the time of application submission, all components of the institution combined must not have received support from the nih totaling more than $6 million per year (in both direct and f&a/indirect costs) in 4 of the last 7 years. for institutions composed of multiple schools and colleges, the $6 million funding limit is based on the amount of nih funding received by all of the schools and colleges within the institution as a whole. note that all activity codes are included in this calculation except the following: c06, s10, and all activity codes starting with a g. a graduate school offers advanced degrees, beyond the undergraduate level, in an academic discipline including m.a., m.s., and ph.d. degrees. health professional schools and colleges are accredited institutions that provide education and training leading to a health professional degree, including but not limited to: b.s.n., m.s.n., d.n.p., m.d., d.d.s., d.o., pharm.d., d.v.m., o.d., d.p.t., d.c., n.d., d.p.m., m.o.t., o.t.d., d.p.t., m.s.-s.l.p., c.sc.d., s.l.p.d., au.d., m.s.p.o., m.s.a.t., and m.p.h. eligible health professional schools/colleges may include schools or colleges of nursing, medicine, dentistry, osteopathy, pharmacy, veterinary medicine, public health, optometry, allied health, chiropractic, naturopathy, podiatry, rehabilitation medicine, physical therapy, orthotics and prosthetics, kinesiology, occupational therapy, and psychology. accreditation must be provided by a body approved for such purpose by the secretary of education. for institutions with multiple campuses, eligibility can be considered for each individual campus (e.g., main, satellite, etc.) only if a unique identifier number and nih ipf number are established for each campus. for institutions that use one identifier number or nih ipf number for all campuses, eligibility is determined for all campuses (e.g., main, satellite, etc.) together. additional eligibility guidance a signed letter is required from the provost or similar official with institution-wide responsibility verifying the eligibility of the applicant institution at the time of application submission according to the eligibility criteria indicated above. see the application instructions for “other attachments” on the sf424(r&r) other project information form in section iv.2 instructions for application submission. final eligibility will be validated by nih prior to award. to assist in determining eligibility, organizations are encouraged to use the nih report website under nih awards by location & organization . a prep application must provide evidence of a subaward to a separate institution , and the grantee may partner with a non-r15-eligible institution. however, applicants should keep the goals of the prep in mind when preparing the application, which include strengthening the research environment of eligible institutions and engaging students from eligible institutions in pain research. it is expected that the project, and two-thirds of the total project budget, will be directed by the pd(s)/pi(s) at r15-eligible institution(s). a letter of support from each collaborator is required verifying the research collaboration at the time of application submission according to the eligibility criteria indicated above. the letter(s) should detail how the proposed research partnership will help to accomplish the proposed pain research project, enhance the r15-eligible institution’s research program, and promote synergy from an integrated, interdisciplinary research partnership(s) among the multiple proposed institutions. see the application instructions for “other attachments” on the sf424(r&r) other project information form in section iv.2 instructions for application submission. foreign organizations non-domestic (non-u.s.) entities (foreign organizations) are not eligible to apply. non-domestic (non-u.s.) components of u.s. organizations are not eligible to apply. foreign components, as defined in the nih grants policy statement , are allowed. required registrations applicant organizations applicant organizations must complete and maintain the following registrations as described in the how to apply - application guide to be eligible to apply for or receive an award. all registrations must be completed prior to the application being submitted. registration can take 6 weeks or more, so applicants should begin the registration process as soon as possible. failure to complete registrations in advance of a due date is not a valid reason for a late submission, please reference nih grants policy statement section 2.3.9.2 electronically submitted applications for additional information system for award management (sam) – applicants must complete and maintain an active registration, which requires renewal at least annually . the renewal process may require as much time as the initial registration. sam registration includes the assignment of a commercial and government entity (cage) code for domestic organizations which have not already been assigned a cage code. nato commercial and government entity (ncage) code – foreign organizations must obtain an ncage code (in lieu of a cage code) in order to register in sam. unique entity identifier (uei) - a uei is issued as part of the sam.gov registration process. the same uei must be used for all registrations, as well as on the grant application. era commons - once the unique organization identifier is established, organizations can register with era commons in tandem with completing their grants.gov registrations; all registrations must be in place by time of submission. era commons requires organizations to identify at least one signing official (so) and at least one program director/principal investigator (pd/pi) account in order to submit an application. grants.gov – applicants must have an active sam registration in order to complete the grants.gov registration. program directors/principal investigators (pd(s)/pi(s)) all pd(s)/pi(s) must have an era commons account. pd(s)/pi(s) should work with their organizational officials to either create a new account or to affiliate their existing account with the applicant organization in era commons. if the pd/pi is also the organizational signing official, they must have two distinct era commons accounts, one for each role. obtaining an era commons account can take up to 2 weeks. eligible individuals (program director/principal investigator) any individual(s) with the skills, knowledge, and resources necessary to carry out the proposed research as the program director(s)/principal investigator(s) (pd(s)/pi(s)) is invited to work with their organization to develop an application for support. individuals from diverse backgrounds, including individuals from underrepresented racial and ethnic groups, individuals with disabilities, and women are always encouraged to apply for nih support. see, reminder: notice of nih's encouragement of applications supporting individuals from underrepresented ethnic and racial groups as well as individuals with disabilities , not-od-22-019 . for institutions/organizations proposing multiple pds/pis, visit the multiple program director/principal investigator policy and submission details in the senior/key person profile (expanded) component of the how to apply - application guide . to be eligible for support under a prep grant, the pd(s)/pi(s) must meet the following additional criteria: each pd/pi must have a primary appointment at either an r15-eligible institution, including professional or graduate schools, undergraduate-focused organizations, or a college within the applicant institution, as defined in “eligible organizations,” above. if proposing multiple pd(s)/pi(s), each pd/pi must be at an r15-eligible institution. each pd/pi may not be the pd/pi of an active nih research grant, including another r15 grant, at the time of award of a prep grant, although they may be one of the key personnel for an active nih grant held by another pd/pi. each pd/pi may not be awarded support under more than one r15 grant at a time, although he or she may have support under successive new or renewal grants. 2. cost sharing.
This NOFO does not require cost sharing as defined in the NIH Grants Policy Statement NIH Grants Policy Statement Section 1.2 Definition of Terms.
3. Additional Information on Eligibility
Number of Applications
Applicant organizations may submit more than one application, provided that each application is scientifically distinct.
The NIH will not accept duplicate or highly overlapping applications under review at the same time, per NIH Grants Policy Statement Section 2.3.7.4 Submission of Resubmission Application . This means that the NIH will not accept:
- A new (A0) application that is submitted before issuance of the summary statement from the review of an overlapping new (A0) or resubmission (A1) application.
- A resubmission (A1) application that is submitted before issuance of the summary statement from the review of the previous new (A0) application.
- An application that has substantial overlap with another application pending appeal of initial peer review (see NIH Grants Policy Statement 2.3.9.4 Similar, Essentially Identical, or Identical Applications ).
Section IV. Application and Submission Information
1. requesting an application package.
The application forms package specific to this opportunity must be accessed through ASSIST, Grants.gov Workspace or an institutional system-to-system solution. Links to apply using ASSIST or Grants.gov Workspace are available in Part 1 of this NOFO. See your administrative office for instructions if you plan to use an institutional system-to-system solution.
2. Content and Form of Application Submission
It is critical that applicants follow the instructions in the Research (R) Instructions in the How to Apply - Application Guide except where instructed in this notice of funding opportunity to do otherwise. Conformance to the requirements in the How to Apply - Application Guide is required and strictly enforced. Applications that are out of compliance with these instructions may be delayed or not accepted for review.
Letter of Intent
Although a letter of intent is not required, is not binding, and does not enter into the review of a subsequent application, the information that it contains allows IC staff to estimate the potential review workload and plan the review.
By the date listed in Part 1. Overview Information , prospective applicants are asked to submit a letter of intent that includes the following information:
- Descriptive title of proposed activity
- Name(s), address(es), and telephone number(s) of the PD(s)/PI(s)
- Names of other key personnel
- Participating institution(s)
- Number and title of this funding opportunity
The letter of intent should be sent to:
Jessica McKlveen, PhD National Center for Complementary & Integrative Health (NCCIH) Telephone: 301-594-8018 Email: [email protected]
Page Limitations
All page limitations described in the How to Apply – Application Guide and the Table of Page Limits must be followed.
The following section supplements the instructions found in the How to Apply – Application Guide and should be used for preparing an application to this NOFO.
SF424(R&R) Cover
All instructions in the How to Apply - Application Guide must be followed.
SF424(R&R) Project/Performance Site Locations
Sf424(r&r) other project information.
Facilities & Other Resources (Required):
- A profile of the scientific background, academic level, and expertise of the students of the applicant institution and any information or estimate of the number who have obtained a health professional baccalaureate or advanced degree and gone on to obtain an academic or professional doctoral or other advanced degree in the health-related sciences during the last 5 years.
- Description of plans to build a broad team of prospective researchers, including students, with a variety of backgrounds, expertise, and skills, and to arrive at major decisions, accounting for different points of view. Personnel from the primary R15-eligible institution(s) should compose a two-thirds majority of the project team .
- Description of the special characteristics of the applicant institution that make it appropriate for an PREP grant awarded through this NOFO to: (1) support the efforts by R15-eligible principal investigators (PIs) at undergraduate-focused institutions or health professional schools and graduate schools to conduct small-scale basic and mechanistic pain research projects; (2) promote integrated, interdisciplinary research partnerships between R15-eligible PIs and additional investigators from U.S. domestic institutions; and (3) enhance the pain research environment at the R15-eligible institution for health professional students or undergraduate and/or graduate students by actively engaging them in the proposed pain research projects.
- Description of the likely impact of a PREP grant on the ability of the PD(s)/PI(s) to engage students in research.
- Description of the likely impact of a PREP grant on the research environment of the applicant institution.
- Description of the likely impact of the PREP grant on the ability of health professional or undergraduate and/or graduate students at the institution to gain experience conducting biomedical research.
- Description of the resources of the grantee institution available for the proposed research (e.g., equipment, supplies, laboratory space, release time, matching funds).
- Although the majority of the research project should be conducted at the R15-eligible institution, the use of special facilities or equipment at another institution is permitted. For any proposed research sites other than the applicant institution, provide a brief description of the resources and access students will need and have to these resources.
Applications without a Facilities & Other Resources document will be withdrawn.
Other Attachments:
Applications that fail to include the following three required ‘other’ attachments will be considered incomplete and will be withdrawn.
1.Team Management Plan (Required three pages maximum):
A key goal of this program is to establish new research partnerships among R15-eligible investigators and other domestic research centers, programs, or institutions with complementary research expertise and/or resources. To ensure that prospective research teams fit the goals of the PREP, a team management plan is required. Applications with team management plans that exceed the three-page limit will be withdrawn.
As an “Other Attachment” entitled Team-Management-Plan.pdf, applications should describe how the research collaborators will function to accomplish program objectives. Team management approaches raised in the subsections listed below should be described in the plan. Note that a “Multiple PD/PI Leadership Plan” may also be submitted as a separate attachment, and if it is included the information in that plan should not be duplicated here. Whereas the Multiple PD/PI Leadership Plan focuses on leadership by and interactions across the PD/PIs, the Team Management Plan focuses on management of the whole team/key personnel. Applicants are encouraged to consult resources to aid in developing effective team-based programs (see e.g., the NCI Collaboration and Team Science Field Guide ).
Organizational structure and team composition: The Team Management Plan should clearly show the organizational structure and composition of the proposed project team. Two-thirds of the proposed research project team should be health professional students trainees or graduate students or undergraduate from the primary R15-eligible institution. The plan should describe a management structure based on project objectives that effectively promote the proposed research. The structure should account for team composition, institutional resources, and policies that conform with PREP objectives outlined in Section I.
Shared leadership, contributions, and distributed responsibility for decision-making: The Team Management Plan should include a description of how the proposed collaborators will work together to direct the overall scientific team to leverage the diverse perspectives, expertise, and skills of the team members to successfully accomplish the goals of the project. One key consideration is that teams employing multidisciplinary approaches and having diverse areas of intellectual and technical expertise are more productive if the process for making decisions incorporates different points of view. The Team Management Plan should describe how major decisions will be made or how conflicts will be resolved.
Resource sharing and allocation across the team: Applications should describe management and decision-making processes that promote collective input for allocation of program resources with flexibility when resources may need to be dynamically reallocated to achieve programmatic goals. A plan for how intra-team, institutional, and regional resources that are integral to the team goals will be shared and made accessible to team members should also be included.
Credit assignment: A plan for how credit and IP will be shared, especially with the R15 institution’s students, should be included. Methods for attributing contributions to publications should be described to enable individual professional assessment in joint projects.
Coordination and communication plans: Practical aspects should be described, including frequency and logistics of real-time communication across all key personnel, consultants, scholars, early-stage investigators etc., and other significant contributors regardless of effort level.
An important and meaningful impact of team science may come from shaping the next generation of pain scientists. Because of the interdisciplinary expertise of the research groups, students are exposed to and can learn a variety of scientific approaches and methodologies, resulting in multifaceted early-stage investigators. Plans for how students trainees will be immersed in and benefit from different approaches taken by the collective team program should be described. This could include shared mentorship, inter-laboratory meetings, all-hands tutorials, shared meeting and document space, inter-laboratory visits, and student presentations.
2. Provost Letter(s) of Support: The application must include a PDF-formatted letter named “ProvostLetter.pdf” (without quotation marks). For MPI applications a signed provost letter is required from each involved institution. The letter must be signed by the provost or similar official with institution-wide responsibility attesting to the following information:
For Undergraduate Focused Institutions:
- The eligible academic component(s) (i.e., the college/school level) must have more undergraduates than graduate students as of the date of submission.
- All the non-health professional components of the institution together have received support from the NIH totaling no more than $6 million per year (in both direct and F&A/indirect costs) in 4 of the last 7 years, as described in Section III, "Eligible Organization".
- Validation that the PD/PI has (or in the case of a multiple PD/PI application that all PD(s)/PI(s) have) a primary appointment at the qualifying component (i.e., the college/school level).
For Health Professional and Graduate Schools:
- The eligible academic component(s) (i.e., the college/school level) must be a health professional or graduate school that awards health professional baccalaureate or advanced degrees in biomedical and/or biobehavioral sciences.
- All components of the institution together have received support from NIH totaling no more than $6 million per year (in both direct and F&A/indirect costs) in 4 of the last 7 years, as described in Section III, “Eligible Organization.”
- Validation that the PD/PI has (or in the case of a multiple PD/PI application that all PD(s)/PI(s) have) a primary appointment at the qualifying component (i.e., the college/school level).
3. Collaborator Letter(s) of Support: Applications must include additional PDF-formatted letter(s) from collaborating subaward holder(s) named “CollaboratorLetter_ Initials .pdf” (without quotation marks). For multiple collaborators, a signed letter is required from each involved collaborator. Note that collaborators do not need to meet the R15-eligibility criteria outlined above. The letter should demonstrate the collaborator's willingness to collaborate with the study lead as well as briefly outline their contributions to the project that will result in a well-integrated, interdisciplinary research approach to the understanding of pain. If the proposed collaboration is a new research partnership among investigators, this information should also be included.
Plan for Enhancing Diverse Perspectives (PEDP)
- In an "Other Attachment" entitled "Plan for Enhancing Diverse Perspectives," all applicants must include a summary of actionable strategies to advance the scientific and technical merit of the proposed project through expanded inclusivity.
- Applicants should align their proposed strategies for PEDP with the research strategy section, providing a holistic and integrated view of how enhancing diverse perspectives and inclusivity are buoyed throughout the application.
- The PEDP will vary depending on the scientific aims, expertise required, the environment and performance site(s), as well as how the project aims are structured.
- Actionable strategies using defined approaches for the inclusion of diverse perspectives in the project;
- Description of how the PEDP will advance the scientific and technical merit of the proposed project;
- Anticipated timeline of proposed PEDP activities;
- Evaluation methods for assessing the progress and success of PEDP activities.
Examples of items that advance inclusivity in research and may be appropriate for a PEDP can include, but are not limited to:
- Partnerships with different types of institutions and organizations (e.g., research-intensive; undergraduate-focused; HBCUs; emerging research institutions; community-based organizations).
- Project frameworks that enable communities and researchers to work collaboratively as equal partners in all phases of the research process.
- Outreach and planned engagement activities to enhance recruitment of individuals from diverse groups as human subjects in clinical trials, including those from underrepresented backgrounds.
- Description of planned partnerships that may enhance geographic and regional diversity.
- Outreach and recruiting activities intended to diversify the pool of applicants for research training programs, such as outreach to prospective applicants from groups underrepresented in the biomedical sciences, for example, individuals from underrepresented racial and ethnic groups, those with disabilities, those from disadvantaged backgrounds, and women.
- Plans to utilize the project infrastructure (i.e., research and structure) to enhance the research environment and support career-advancing opportunities for junior, early- and mid-career researchers.
- Transdisciplinary research projects and collaborations among researchers from fields beyond the biological sciences, such as physics, engineering, mathematics, computational biology, computer and data sciences, as well as bioethics.
Examples of items that are not appropriate in a PEDP include, but are not limited to:
- Selection or hiring of personnel for a research team based on their race, ethnicity, or sex (including gender identify, sexual orientation, or transgender status).
- A training or mentorship program limited to certain researchers based on their race, ethnicity, or sex (including gender identify, sexual orientation, or transgender status).
For further information on the Plan for Enhancing Diverse Perspectives (PEDP), please see PEDP guidance materials .
SF424(R&R) Senior/Key Person Profile
R&r or modular budget.
- The total budget for all years of the proposed project must be requested in Budget Period 1. Do not complete Budget Periods 2 or 3. They are not required and will not be accepted with the application.
- Applicants submitting an application with direct costs of $250,000 or less (total for all years, excluding consortium Facilities and Administrative [F&A] costs) must use the Modular Budget.
- Applicants submitting an application with direct costs of $250,001 - $375,000 (total for all years, excluding consortium Facilities and Administrative [F&A] costs) must use the R&R Budget.
- Students must be compensated for their participation in the laboratory's research and in accord with institutional policies. Student salaries can be requested in the R15 budget, or other resources at the university can be used to pay them for their participation. Undergraduate students who are compensated from the R15 grant or other institutional funds should receive at least the national minimum wage . Compensation through course credit hours towards graduation is allowable, but must be justified. If universities/colleges provide room and board for summer research students, details must be provided in the application.
- NIH does not fund stipends for undergraduates on R15 awards.
Budget Justification:
Personnel Justification: Since a primary objective of the PREP is to expose and incorporate students into multidisciplinary pain research, PD(s)/PI(s) must include health professional or undergraduate and/or graduate students from the applicant institution/applicant component in the proposed research. Students from the R15-eligible institution should compose the majority of the research team (two thirds or more). Indicate aspects of the proposed research in which students will participate. If participating students have not yet been identified, the number and academic level of those to be involved should be provided. Collaborators or consultants for the project should provide additional budget information, including their names, their organizational affiliations, and the services they will perform.
PEDP implementation costs: Applicants may include allowable costs associated with PEDP implementation (as outlined in the Grants Policy Statement section 7): https://grants.nih.gov/grants/policy/nihgps/html5/section_7/7.1_general.htm.
R&R Subaward Budget
Phs 398 cover page supplement, phs 398 research plan.
All instructions in the How to Apply - Application Guide must be followed, with the following additional instructions:
Research Strategy:
The research strategy must address how the proposed project intends to accomplish all three objectives of this program, including: 1) Supporting the efforts by R15-eligible principal investigators (PIs) at undergraduate-focused institutions OR health professional schools and graduate schools to conduct small-scale basic and mechanistic pain research; (2) promoting integrated, interdisciplinary research partnerships between R15-eligible PIs and additional investigators from U.S. domestic institutions; and (3) enhancing the pain research environment at the R15-eligible institution for health professional students or undergraduate and/or graduate students by actively engaging them in the proposed pain research projects.
Applications should include a detailed description of a research approach that will produce rigorous data that can be disseminated and advance our basic and mechanistic understanding of pain. Additionally, the research strategy should detail how the proposed research partnership includes sufficient integrative pain expertise and related resources and/or institutional infrastructure that increase the likelihood of success. The application should detail how the proposed scientific research and proposed program and research partnership will have a substantial effect on strengthening the research environment at the proposed applicant’s institution.
Applications should provide details on how the research project will be directed by the R15-eligible PI and how two-thirds of the research project will be conducted at the R15-eligible institution. The research strategy should detail how the research team will recruit additional prospective investigators, including students, from a range of backgrounds, skills, and expertise for the broad pool of researchers who may apply to participate and contribute to the project. Applications should include details about how the investigators will cooperate and coordinate their activities with other HEAL investigators at PD/PI meetings, including (but not limited to) other investigators in the R15 program, the HEAL Annual Scientific and PURPOSE meetings.Proposed PD/PI(s) should include evidence of experience supervising students in previous research efforts, as well as describing any innovative approaches to engage students in the proposed pain research project. Applications should provide additional details outlining student involvement in the research project by addressing the following questions:
- How will students engage in conducting hands-on rigorous research?
- How will students participate in research activities such as planning, execution, and/or analysis of the research?
- Are there any additional plans for student involvement, such as presentation at local or national meetings, participation in publication of research findings, and development of, or participation in, collaborative activities?
- How will the project provide students with adequate opportunities to improve their research capabilities and support their progress toward a future career in pain research?
- Note-The purpose of this program is to support pain research projects, not student training. Formal training plans (e.g., non-research activities, didactic training, seminars) should not be provided, although a brief description of activities related to enhancing students’ research capabilities and progress (e.g., the use of individual development plans) is permitted. Furthermore, applications should not include independent student research projects.
Resource Sharing Plan : Individuals are required to comply with the instructions for the Resource Sharing Plans as provided in the How to Apply - Application Guide .
Other Plan(s):
All instructions in the How to Apply - Application Guide must be followed, with the following additional instructions:
- All applicants planning research (funded or conducted in whole or in part by NIH) that results in the generation of scientific data are required to comply with the instructions for the Data Management and Sharing Plan. All applications, regardless of the amount of direct costs requested for any one year, must address a Data Management and Sharing Plan.
The NIH HEAL Initiative has additional requirements that must be addressed in the Data Management and Sharing Plan. All HEAL-generated data must be shared through the HEAL Initiative Data Ecosystem following HEAL’s compliance guidance ( https://heal.nih.gov/data/complying-heal-data-sharing-policy ). Specifically, HEAL applicants must include:
- Plans to submit data and metadata (and code, if applicable) to a HEAL-compliant data repository ( https://www.healdatafair.org/resources/guidance/selection ) and follow requirements of the selected repository.
- Plans to register your study with the HEAL platform within one year of award ( https://heal.github.io/platform-documentation/study-registration/ ).
- Plans to submit HEAL-defined study-level metadata within one year of award (HTTP ://github.com/HEAL/heal-metadata-schemas/blob/main/for-investigators-how-to/study-level-metadata-fields/study-metadata-schema-for-humans.pdf ) and https://heal.github.io/platform-documentation/slmd_submission/ .
- Plans to submit data dictionaries to the HEAL Data Ecosystem, if applicable.
- HEAL pain clinical studies must include a plan to use HEAL core Common Data Elements (CDEs) ( https://heal.nih.gov/data/common-data-elements ). NIH HEAL Initiative clinical studies that are using copyrighted questionaries are required to obtain licenses for use prior to initiating data collection. Licenses must be shared with the HEAL CDE team and the program officer prior to use of copyrighted materials.
- To the extent possible, all other (nonpain) HEAL studies conducting clinical trials or research involving human subjects are expected to use questionnaires by the HEAL Common Data Elements (CDE) Program ( https://heal.nih.gov/data/common-data-elements ) if applicable and relevant to their research.
- Studies using CDEs, regardless of whether they are part of the HEAL repository, will be required to report which questionnaires are being used.
- To the extent possible, NIH HEAL Initiative awardees are expected to integrate broad data sharing consent language into their informed consent forms.
The NIH HEAL Initiative has developed additional details and resources to fulfill these requirements ( https://www.healdatafair.org/resources/road-map ). Budgeting guidance for data sharing can be found in NOT-OD-21-015 and the NIH Scientific Data Sharing site .
Appendix: Only limited Appendix materials are allowed. Follow all instructions for the Appendix as described in the How to Apply - Application Guide .
- No publications or other material, with the exception of blank questionnaires or blank surveys, may be included in the Appendix.
PHS Human Subjects and Clinical Trials Information
When involving human subjects research, clinical research, and/or NIH-defined clinical trials (and when applicable, clinical trials research experience) follow all instructions for the PHS Human Subjects and Clinical Trials Information form in the How to Apply - Application Guide , with the following additional instructions:
If you answered “Yes” to the question “Are Human Subjects Involved?” on the R&R Other Project Information form, you must include at least one human subjects study record using the Study Record: PHS Human Subjects and Clinical Trials Information form or Delayed Onset Study record.
Study Record: PHS Human Subjects and Clinical Trials Information
Section 2 - Study Population Characteristics
2.5 Recruitment and Retention Plan
Describe the following:
- Recruitment milestones;
- The planned recruitment methods, including use of contact lists (participants and/or sites), databases or other pre-screening resources, advertisements, outreach, media / social media and referral networks or groups;
- If there are known participant or study-related barriers to accrual or participation (based on literature or prior experience), please list these barriers and describe plans to address them to optimize success;
- Contingency plans for participant accrual if enrollment significantly lags behind accrual benchmarks; 5) participant retention and adherence strategies; and 6) possible competition from other trials for study participants.
2.7 Study Timeline
Include a table or graph of the overall study timeline. This is expected to be a visual representation (such as a Gantt chart) of recruitment milestones and key project management activities. A narrative is not expected in this section.
The study timeline should include recruitment milestones that need to be met throughout the life cycle of the clinical trial to ensure its success, and the subtasks that will be used to reach the recruitment milestones. In the timeline, the study duration is expected to be displayed in months. The timeline should include, but is not limited to, the following:
(a) When the study opens to enrollment (b) When recruitment milestones (see below) are met (c) What subtasks are needed to reach of the recruitment milestones (d) When final transfer of the data will occur (e) When analysis of the study data will occur (f) When the primary study manuscript will be submitted for publication
Delayed Onset Study
Note: Delayed onset does NOT apply to a study that can be described but will not start immediately (i.e., delayed start). All instructions in the How to Apply - Application Guide must be followed.
PHS Assignment Request Form
3. unique entity identifier and system for award management (sam).
See Part 2. Section III.1 for information regarding the requirement for obtaining a unique entity identifier and for completing and maintaining active registrations in System for Award Management (SAM), NATO Commercial and Government Entity (NCAGE) Code (if applicable), eRA Commons, and Grants.gov
4. Submission Dates and Times
Part I. contains information about Key Dates and times. Applicants are encouraged to submit applications before the due date to ensure they have time to make any application corrections that might be necessary for successful submission. When a submission date falls on a weekend or Federal holiday , the application deadline is automatically extended to the next business day.
Organizations must submit applications to Grants.gov (the online portal to find and apply for grants across all Federal agencies). Applicants must then complete the submission process by tracking the status of the application in the eRA Commons , NIH’s electronic system for grants administration. NIH and Grants.gov systems check the application against many of the application instructions upon submission. Errors must be corrected and a changed/corrected application must be submitted to Grants.gov on or before the application due date and time. If a Changed/Corrected application is submitted after the deadline, the application will be considered late. Applications that miss the due date and time are subjected to the NIH Grants Policy Statement Section 2.3.9.2 Electronically Submitted Applications .
Applicants are responsible for viewing their application before the due date in the eRA Commons to ensure accurate and successful submission.
Information on the submission process and a definition of on-time submission are provided in the How to Apply – Application Guide .
5. Intergovernmental Review (E.O. 12372)
This initiative is not subject to intergovernmental review.
6. Funding Restrictions
All NIH awards are subject to the terms and conditions, cost principles, and other considerations described in the NIH Grants Policy Statement .
Pre-award costs are allowable only as described in the NIH Grants Policy Statement Section 7.9.1 Selected Items of Cost .
Applications must be submitted electronically following the instructions described in the How to Apply - Application Guide . Paper applications will not be accepted.
Applicants must complete all required registrations before the application due date. Section III. Eligibility Information contains information about registration.
For assistance with your electronic application or for more information on the electronic submission process, visit How to Apply – Application Guide . If you encounter a system issue beyond your control that threatens your ability to complete the submission process on-time, you must follow the Dealing with System Issues guidance. For assistance with application submission, contact the Application Submission Contacts in Section VII .
Important reminders:
All PD(s)/PI(s) must include their eRA Commons ID in the Credential field of the Senior/Key Person Profile form . Failure to register in the Commons and to include a valid PD/PI Commons ID in the credential field will prevent the successful submission of an electronic application to NIH. See Section III of this NOFO for information on registration requirements.
The applicant organization must ensure that the unique entity identifier provided on the application is the same identifier used in the organization’s profile in the eRA Commons and for the System for Award Management. Additional information may be found in the How to Apply - Application Guide .
See more tips for avoiding common errors.
Applications must include a PEDP submitted as Other Project Information as an attachment. Applications that fail to include a PEDP will be considered incomplete and will be administratively withdrawn before review.
Upon receipt, applications will be evaluated for completeness and compliance with application instructions by the Center for Scientific Review and responsiveness by components of participating organizations , NIH. Applications that are incomplete, non-compliant and/or nonresponsive will not be reviewed.
In order to expedite review, applicants are requested to notify the NCCIH Referral Office by email at [email protected] when the application has been submitted. Please include the NOFO and title, PD/PI name, and title of the application.
Recipients or subrecipients must submit any information related to violations of federal criminal law involving fraud, bribery, or gratuity violations potentially affecting the federal award. See Mandatory Disclosures, 2 CFR 200.113 and NIH Grants Policy Statement Section 4.1.35 .
Send written disclosures to the NIH Chief Grants Management Officer listed on the Notice of Award for the IC that funded the award and to the HHS Office of Inspector Grant Self Disclosure Program at [email protected] .
Post Submission Materials
Applicants are required to follow the instructions for post-submission materials, as described in the policy
The following post-submission materials will be accepted: Team Management Plan (e.g., due to the hiring, replacement, or loss of an investigator).
Section V. Application Review Information
1. criteria.
Only the review criteria described below will be considered in the review process. Applications submitted to the NIH in support of the NIH mission are evaluated for scientific and technical merit through the NIH peer review system.
For this particular NOFO, note the following:
The purpose of this HEAL Initiative program is to (1) support the efforts by R15-eligible principal investigators (PIs) at primarily undergraduate-focused serving institutions or health professional schools and graduate schools to conduct small-scale basic and mechanistic pain research projects ; (2) promote integrated, interdisciplinary research partnerships between R15-eligible PIs and investigators from U.S. domestic institutions; and (3) enhance the pain research environment at the R15-eligible institution for health professional students or undergraduate and/or graduate students by actively engaging them in the proposed pain research projects.
Applications in response to this notice of funding opportunity (NOFO) should include plans to accomplish these goals. Specifically, applications should include a rigorous plan for conducting basic and mechanistic pain research projects in the Research Strategy section of the application . In addition, a research partnership between the PI’s institution and at least one investigator from a separate U.S. domestic institution that provides resources and/or expertise that will enhance the proposed pain research program must be included in a separate Team Management Plan. The proposed partnership will be a sub-award agreement(s) with at least one partnering institution, which does not need to be R15-eligible. The budget of all sub-awards must not exceed one third of the total budget. Furthermore, applications must include a Facilities & Other Resources document that demonstrates active involvement of health professional students or undergraduate and/or graduate students from the R15-eligible institution(s) in the proposed pain research projects.
Although preliminary data are not required for an R15 application, they may be included if available. The scientific foundation for the proposed research should be based on published research and/or any available preliminary data.
A proposed Clinical Trial application may include study design, methods, and intervention that are not by themselves innovative but address important questions or unmet needs. Additionally, the results of the clinical trial may indicate that further clinical development of the intervention is unwarranted or lead to new avenues of scientific investigation.
Reviewers will provide an overall impact score to reflect their assessment of the likelihood for the project to exert a sustained, powerful influence on the research field(s) involved, in consideration of the following review criteria and additional review criteria (as applicable for the project proposed).As part of the overall impact score, reviewers should consider and indicate how the Plan for Enhancing Diverse Perspectives affects the scientific merit of the project.
Reviewers will consider each of the review criteria below in the determination of scientific merit and give a separate score for each. An application does not need to be strong in all categories to be judged likely to have major scientific impact. For example, a project that by its nature is not innovative may be essential to advance a field.
Does the project address an important problem or a critical barrier to progress in the field? Is the prior research that serves as the key support for the proposed project rigorous? If the aims of the project are achieved, how will scientific knowledge, technical capability, and/or clinical practice be improved? How will successful completion of the aims change the concepts, methods, technologies, treatments, services, or preventative interventions that drive this field?
In addition, for applications involving clinical trials
Are the scientific rationale and need for a clinical trial to test the proposed hypothesis or intervention well supported by preliminary data, clinical and/or preclinical studies, or information in the literature or knowledge of biological mechanisms? For trials focusing on clinical or public health endpoints, is this clinical trial necessary for testing the safety, efficacy or effectiveness of an intervention that could lead to a change in clinical practice, community behaviors or health care policy? For trials focusing on mechanistic, behavioral, physiological, biochemical, or other biomedical endpoints, is this trial needed to advance scientific understanding?
Specific to this NOFO:
Taking into consideration the type of R15-eligible institution the application has been submitted from, if funded, will this grant have a substantial effect on strengthening the research environment at the applicant institution and exposing students to research ?
Does the project adequately describe how the research partnership will advance our understanding of pain conditions?
If the aims of the project are achieved, will the project yield rigorous data that can be disseminated and is likely to be important to the field?
Will the proposed collaboration appropriately improve the R15 institutional environment in a manner to support more students to engage in pain research at that institution?
Are the PD(s)/PI(s), collaborators, and other researchers well suited to the project? If Early Stage Investigators or those in the early stages of independent careers, do they have appropriate experience and training? If established, have they demonstrated an ongoing record of accomplishments that have advanced their field(s)? If the project is collaborative or multi-PD/PI, do the investigators have complementary and integrated expertise; are their leadership approach, governance and organizational structure appropriate for the project?
With regard to the proposed leadership for the project, do the PD/PI(s) and key personnel have the expertise, experience, and ability to organize, manage and implement the proposed clinical trial and meet milestones and timelines? Do they have appropriate expertise in study coordination, data management and statistics? For a multicenter trial, is the organizational structure appropriate and does the application identify a core of potential center investigators and staffing for a coordinating center?
Does the application provide details about how the research project will be directed by the R15-eligible PI and how two-thirds of the research project will be conducted at the R15-eligible institution?
Is it clear how the applicant intends to recruit additional prospective investigators, including students, from a range of backgrounds, skills, and expertise for the pool of researchers who may apply to address the proposed scientific problem?
Will the combined scientific expertise (of the proposed collaborative research team) likely result in a well-integrated, interdisciplinary research approach to the understanding of pain?
Does the team of investigators include sufficient integrative pain expertise for the proposed research?
How appropriate is the PD/PI’'s experience in supervising and engaging students in research?
Does the application include details about how the investigators will cooperate and coordinate their activities with other HEAL investigators at PD/PI meetings, including (but not limited to) other investigators in the R15 program, the HEAL Annual Scientific and PURPOSE meetings?
Team Management Plan (Attachment):
How fair and adequate are the governance processes for decision making, conflict resolution, and resource allocation outlined in the plan?
How effective is the plan for team leadership and management with sufficient examples of distributed responsibility?
How well would the program leadership create a sustainable environment for maintaining cohesiveness, productivity, and shared vision?
How adequate are the management plans for shared professional credit?
If shared research resources will be utilized, how adequate are the plans for resource sharing and allocation to ensure that all team members will have the access they require?
How well does the plan include examples of team coordination and communication?
How clearly does the plan include details about which personnel are available at the R15-eligible institution(s), including health professional students or graduate students or undergraduate students, that would compose a two-thirds majority of the project team and how they would contribute to the research project?
How well does the management plan outline how the collaborative partnership will supervise and engage students?
Does the application challenge and seek to shift current research or clinical practice paradigms by utilizing novel theoretical concepts, approaches or methodologies, instrumentation, or interventions? Are the concepts, approaches or methodologies, instrumentation, or interventions novel to one field of research or novel in a broad sense? Is a refinement, improvement, or new application of theoretical concepts, approaches or methodologies, instrumentation, or interventions proposed?
Does the design/research plan include innovative elements, as appropriate, that enhance its sensitivity, potential for information or potential to advance scientific knowledge or clinical practice?
Does the proposed research include innovative interdisciplinary pain research topics?
Is the proposed research partnership a new collaboration between investigators?
Are innovative approaches for engaging health professional or undergraduate and/or graduate students in research proposed?
Are the overall strategy, methodology, and analyses well-reasoned and appropriate to accomplish the specific aims of the project? Have the investigators included plans to address weaknesses in the rigor of prior research that serves as the key support for the proposed project? Have the investigators presented strategies to ensure a robust and unbiased approach, as appropriate for the work proposed? Are potential problems, alternative strategies, and benchmarks for success presented? If the project is in the early stages of development, will the strategy establish feasibility and will particularly risky aspects be managed? Have the investigators presented adequate plans to address relevant biological variables, such as sex, for studies in vertebrate animals or human subjects?
If the project involves human subjects and/or NIH-defined clinical research, are the plans to address 1) the protection of human subjects from research risks, and 2) inclusion (or exclusion) of individuals on the basis of sex/gender, race, and ethnicity, as well as the inclusion or exclusion of individuals of all ages (including children and older adults), justified in terms of the scientific goals and research strategy proposed?
Does the application adequately address the following, if applicable
Study Design
Is the study design justified and appropriate to address primary and secondary outcome variable(s)/endpoints that will be clear, informative and relevant to the hypothesis being tested? Is the scientific rationale/premise of the study based on previously well-designed preclinical and/or clinical research? Given the methods used to assign participants and deliver interventions, is the study design adequately powered to answer the research question(s), test the proposed hypothesis/hypotheses, and provide interpretable results? Is the trial appropriately designed to conduct the research efficiently? Are the study populations (size, gender, age, demographic group), proposed intervention arms/dose, and duration of the trial, appropriate and well justified?
Are potential ethical issues adequately addressed? Is the process for obtaining informed consent or assent appropriate? Is the eligible population available? Are the plans for recruitment outreach, enrollment, retention, handling dropouts, missed visits, and losses to follow-up appropriate to ensure robust data collection? Are the planned recruitment timelines feasible and is the plan to monitor accrual adequate? Has the need for randomization (or not), masking (if appropriate), controls, and inclusion/exclusion criteria been addressed? Are differences addressed, if applicable, in the intervention effect due to sex/gender and race/ethnicity?
Are the plans to standardize, assure quality of, and monitor adherence to, the trial protocol and data collection or distribution guidelines appropriate? Is there a plan to obtain required study agent(s)? Does the application propose to use existing available resources, as applicable?
Data Management and Statistical Analysis
Are planned analyses and statistical approach appropriate for the proposed study design and methods used to assign participants and deliver interventions? Are the procedures for data management and quality control of data adequate at clinical site(s) or at center laboratories, as applicable? Have the methods for standardization of procedures for data management to assess the effect of the intervention and quality control been addressed? Is there a plan to complete data analysis within the proposed period of the award?
Taking into consideration the type of R15-eligible institution the application has been submitted from, how suitable are the plans for ensuring that students are well integrated into the research program?
How will this project provide students with a high-quality research experience focused on the execution, analysis, and reporting of the study?
Would students have adequate opportunities to present at national or local meetings, publish research findings, and/or participate in other collaborative activities?
Would the proposed research project provide adequate opportunities for students to improve their research capabilities and support their progress toward a biomedical research career?
Will the scientific environment in which the work will be done contribute to the probability of success? Are the institutional support, equipment and other physical resources available to the investigators adequate for the project proposed? Will the project benefit from unique features of the scientific environment, subject populations, or collaborative arrangements?
If proposed, are the administrative, data coordinating, enrollment and laboratory/testing centers, appropriate for the trial proposed?
Does the application adequately address the capability and ability to conduct the trial at the proposed site(s) or centers? Are the plans to add or drop enrollment centers, as needed, appropriate?
If international site(s) is/are proposed, does the application adequately address the complexity of executing the clinical trial?
If multi-sites/centers, is there evidence of the ability of the individual site or center to: (1) enroll the proposed numbers; (2) adhere to the protocol; (3) collect and transmit data in an accurate and timely fashion; and, (4) operate within the proposed organizational structure?
Does the "Facilities & Other Resources" attachment describe strong and innovative approaches to how students or trainees will participate in the research project?
Does the application demonstrate appropriate plans to recruit health professional or undergraduate and/or graduate students from diverse backgrounds to participate in the research project?
Does the application provide a plan to aid students at the R15-eligible institution/academic component to pursue careers in the biomedical sciences?
Do(es) the PD/PI(s) have sufficient time and institutional support to conduct the proposed project?
Is there synergy to be gained from the integrated, interdisciplinary research partnership(s) among the multiple proposed institutions?
As applicable for the project proposed, reviewers will evaluate the following additional items while determining scientific and technical merit, and in providing an overall impact score, but will not give separate scores for these items.
Specific to applications involving clinical trials
Is the study timeline described in detail, taking into account start-up activities, the anticipated rate of enrollment, and planned follow-up assessment? Is the projected timeline feasible and well justified? Does the project incorporate efficiencies and utilize existing resources (e.g., CTSAs, practice-based research networks, electronic medical records, administrative database, or patient registries) to increase the efficiency of participant enrollment and data collection, as appropriate?
Are potential challenges and corresponding solutions discussed (e.g., strategies that can be implemented in the event of enrollment shortfalls)?
Specific to this NOFO: Are the clinical trial recruitment milestones feasible given the proposed study timeline?
For research that involves human subjects but does not involve one of the categories of research that are exempt under 45 CFR Part 46, the committee will evaluate the justification for involvement of human subjects and the proposed protections from research risk relating to their participation according to the following five review criteria: 1) risk to subjects, 2) adequacy of protection against risks, 3) potential benefits to the subjects and others, 4) importance of the knowledge to be gained, and 5) data and safety monitoring for clinical trials.
For research that involves human subjects and meets the criteria for one or more of the categories of research that are exempt under 45 CFR Part 46, the committee will evaluate: 1) the justification for the exemption, 2) human subjects involvement and characteristics, and 3) sources of materials. For additional information on review of the Human Subjects section, please refer to the Guidelines for the Review of Human Subjects .
When the proposed project involves human subjects and/or NIH-defined clinical research, the committee will evaluate the proposed plans for the inclusion (or exclusion) of individuals on the basis of sex/gender, race, and ethnicity, as well as the inclusion (or exclusion) of individuals of all ages (including children and older adults) to determine if it is justified in terms of the scientific goals and research strategy proposed. For additional information on review of the Inclusion section, please refer to the Guidelines for the Review of Inclusion in Clinical Research .
The committee will evaluate the involvement of live vertebrate animals as part of the scientific assessment according to the following three points: (1) a complete description of all proposed procedures including the species, strains, ages, sex, and total numbers of animals to be used; (2) justifications that the species is appropriate for the proposed research and why the research goals cannot be accomplished using an alternative non-animal model; and (3) interventions including analgesia, anesthesia, sedation, palliative care, and humane endpoints that will be used to limit any unavoidable discomfort, distress, pain and injury in the conduct of scientifically valuable research. Methods of euthanasia and justification for selected methods, if NOT consistent with the AVMA Guidelines for the Euthanasia of Animals, is also required but is found in a separate section of the application. For additional information on review of the Vertebrate Animals Section, please refer to the Worksheet for Review of the Vertebrate Animals Section.
Reviewers will assess whether materials or procedures proposed are potentially hazardous to research personnel and/or the environment, and if needed, determine whether adequate protection is proposed.
For Resubmissions, the committee will evaluate the application as now presented, taking into consideration the responses to comments from the previous scientific review group and changes made to the project.
Not applicable.
Not applicable.
As applicable for the project proposed, reviewers will consider each of the following items, but will not give scores for these items, and should not consider them in providing an overall impact score.
Reviewers will assess whether the project presents special opportunities for furthering research programs through the use of unusual talent, resources, populations, or environmental conditions that exist in other countries and either are not readily available in the United States or augment existing U.S. resources.
Reviewers will assess the information provided in this section of the application, including 1) the Select Agent(s) to be used in the proposed research, 2) the registration status of all entities where Select Agent(s) will be used, 3) the procedures that will be used to monitor possession use and transfer of Select Agent(s), and 4) plans for appropriate biosafety, biocontainment, and security of the Select Agent(s).
Reviewers will comment on whether the Resource Sharing Plan(s) (e.g., Sharing Model Organisms ) or the rationale for not sharing the resources, is reasonable.
For projects involving key biological and/or chemical resources, reviewers will comment on the brief plans proposed for identifying and ensuring the validity of those resources.
Reviewers will consider whether the budget and the requested period of support are fully justified and reasonable in relation to the proposed research.
2. Review and Selection Process Applications will be evaluated for scientific and technical merit by (an) appropriate Scientific Review Group(s) convened by NCCIH, in accordance with NIH peer review policies and practices , using the stated review criteria. Assignment to a Scientific Review Group will be shown in the eRA Commons. As part of the scientific peer review, all applications will receive a written critique. Applications may undergo a selection process in which only those applications deemed to have the highest scientific and technical merit (generally the top half of applications under review) will be discussed and assigned an overall impact score. Appeals of initial peer review will not be accepted for applications submitted in response to this NOFO. Applications will be assigned on the basis of established PHS referral guidelines to the appropriate NIH Institute or Center. Applications will compete for available funds with all other recommended applications submitted in response to this NOFO. Following initial peer review, recommended applications will receive a second level of review by the appropriate national Advisory Council or Board. The following will be considered in making funding decisions: Scientific and technical merit of the proposed project, including the PEDP, as determined by scientific peer review Availability of funds. Relevance of the proposed project to program priorities. Please note that reviewers will not consider race, ethnicity, age, or sex (including gender identity, sexual orientation or transgender status) of a researcher, award participant, or trainee, even in part, in providing critiques, scores, or funding recommendations. NIH will not consider such factors in making its funding decisions. If the application is under consideration for funding, NIH will request "just-in-time" information from the applicant as described in the NIH Grants Policy Statement Section 2.5.1. Just-in-Time Procedures . This request is not a Notice of Award nor should it be construed to be an indicator of possible funding. Prior to making an award, NIH reviews an applicant’s federal award history in SAM.gov to ensure sound business practices. An applicant can review and comment on any information in the Responsibility/Qualification records available in SAM.gov. NIH will consider any comments by the applicant in the Responsibility/Qualification records in SAM.gov to ascertain the applicant’s integrity, business ethics, and performance record of managing Federal awards per 2 CFR Part 200.206 “Federal awarding agency review of risk posed by applicants.” This provision will apply to all NIH grants and cooperative agreements except fellowships. 3. Anticipated Announcement and Award Dates
After the peer review of the application is completed, the PD/PI will be able to access his or her Summary Statement (written critique) via the eRA Commons . Refer to Part 1 for dates for peer review, advisory council review, and earliest start date.
Information regarding the disposition of applications is available in the NIH Grants Policy Statement Section 2.4.4 Disposition of Applications .
Section VI. Award Administration Information
1. award notices.
A Notice of Award (NoA) is the official authorizing document notifying the applicant that an award has been made and that funds may be requested from the designated HHS payment system or office. The NoA is signed by the Grants Management Officer and emailed to the recipient’s business official.
In accepting the award, the recipient agrees that any activities under the award are subject to all provisions currently in effect or implemented during the period of the award, other Department regulations and policies in effect at the time of the award, and applicable statutory provisions.
Recipients must comply with any funding restrictions described in Section IV.6. Funding Restrictions . Any pre-award costs incurred before receipt of the NoA are at the applicant's own risk. For more information on the Notice of Award, please refer to the NIH Grants Policy Statement Section 5. The Notice of Award and NIH Grants & Funding website, see Award Process.
Individual awards are based on the application submitted to, and as approved by, the NIH and are subject to the IC-specific terms and conditions identified in the NoA.
ClinicalTrials.gov: If an award provides for one or more clinical trials. By law (Title VIII, Section 801 of Public Law 110-85), the "responsible party" must register and submit results information for certain “applicable clinical trials” on the ClinicalTrials.gov Protocol Registration and Results System Information Website ( https://register.clinicaltrials.gov ). NIH expects registration and results reporting of all trials whether required under the law or not. For more information, see https://grants.nih.gov/policy/clinical-trials/reporting/index.htm
Institutional Review Board or Independent Ethics Committee Approval: Recipient institutions must ensure that all protocols are reviewed by their IRB or IEC. To help ensure the safety of participants enrolled in NIH-funded studies, the recipient must provide NIH copies of documents related to all major changes in the status of ongoing protocols.
Data and Safety Monitoring Requirements: The NIH policy for data and safety monitoring requires oversight and monitoring of all NIH-conducted or -supported human biomedical and behavioral intervention studies (clinical trials) to ensure the safety of participants and the validity and integrity of the data. Further information concerning these requirements is found at http://grants.nih.gov/grants/policy/hs/data_safety.htm and in the application instructions (SF424 (R&R) and PHS 398).
Investigational New Drug or Investigational Device Exemption Requirements: Consistent with federal regulations, clinical research projects involving the use of investigational therapeutics, vaccines, or other medical interventions (including licensed products and devices for a purpose other than that for which they were licensed) in humans under a research protocol must be performed under a Food and Drug Administration (FDA) investigational new drug (IND) or investigational device exemption (IDE).
2. Administrative and National Policy Requirements
The following Federal wide and HHS-specific policy requirements apply to awards funded through NIH:
- The rules listed at 2 CFR Part 200 , Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards.
- All NIH grant and cooperative agreement awards include the NIH Grants Policy Statement as part of the terms and conditions in the Notice of Award (NoA). The NoA includes the requirements of this NOFO. For these terms of award, see the NIH Grants Policy Statement Part II: Terms and Conditions of NIH Grant Awards, Subpart A: General and Part II: Terms and Conditions of NIH Grant Awards, Subpart B: Terms and Conditions for Specific Types of Grants, Recipients, and Activities .
- HHS recognizes that NIH research projects are often limited in scope for many reasons that are nondiscriminatory, such as the principal investigator’s scientific interest, funding limitations, recruitment requirements, and other considerations. Thus, criteria in research protocols that target or exclude certain populations are warranted where nondiscriminatory justifications establish that such criteria are appropriate with respect to the health or safety of the subjects, the scientific study design, or the purpose of the research. For additional guidance regarding how the provisions apply to NIH grant programs, please contact the Scientific/Research Contact that is identified in Section VII under Agency Contacts of this NOFO.
All federal statutes and regulations relevant to federal financial assistance, including those highlighted in NIH Grants Policy Statement Section 4 Public Policy Requirements, Objectives and Other Appropriation Mandates.
Recipients are responsible for ensuring that their activities comply with all applicable federal regulations. NIH may terminate awards under certain circumstances. See 2 CFR Part 200.340 Termination and NIH Grants Policy Statement Section 8.5.2 Remedies for Noncompliance or Enforcement Actions: Suspension, Termination, and Withholding of Support .
3. Data Management and Sharing
Consistent with the 2023 NIH Policy for Data Management and Sharing, when data management and sharing is applicable to the award, recipients will be required to adhere to the Data Management and Sharing requirements as outlined in the NIH Grants Policy Statement . Upon the approval of a Data Management and Sharing Plan, it is required for recipients to implement the plan as described.
HEAL Data Sharing Requirements
NIH intends to maximize the impact of NIH HEAL Initiative-supported projects through broad and rapid data sharing. All NIH HEAL Initiative award recipients, regardless of the amount of direct costs requested for any one year, are required to comply with the HEAL Public Access and Data Sharing Policy. NIH HEAL Initiative award recipients must follow all requirements and timelines developed through the HEAL Initiative Data Ecosystem ( https://heal.nih.gov/about/heal-data-ecosystem ), as described in the initiative’s compliance guidance (See “Already Funded” section: https://heal.nih.gov/data/complying-heal-data-sharing-policy ):
1. Select a HEAL-compliant data repository ( https://www.healdatafair.org/resources/guidance/selection )
- Data generated by NIH HEAL Initiative-funded projects must be submitted to study-appropriate, HEAL-compliant data repositories to ensure the data is accessible via the HEAL Initiative Data Ecosystem.
- Some repositories require use of specific data dictionaries or structured data elements, so knowing your repository’s requirements up front can help reduce the burden of preparing data for submission.
- HEAL-funded awardees must follow requirements for selected repository.
2. Within one year of award, register your study with the HEAL platform ( https://heal.github.io/platform-documentation/study-registration/ )
- This process will connect the platform to information about your study and data, including metadata, and identify the selected repository. HEAL requests initial submission within one year of award, with annual updates, and to be updated in accordance with any release of study data.
3. Within one year of award, submit HEAL-specific study-level metadata.
- Some of the required study-level metadata ( https://github.com/HEAL/heal-metadata-schemas/blob/main/for-investigators-how-to/study-level-metadata-fields/study-metadata-schema-for-humans.pdf ) will be autopopulated as part of the registration process.
4. Submit data and metadata (and code, if applicable) to HEAL-compliant repository
- At the completion of the study and/or when prepared to make the final data deposits in the repositor(ies) of choice, ensure your study registration ( https://heal.github.io/platform-documentation/study-registration/ ) is complete.
- Submit data dictionaries to the HEAL data ecosystem, if applicable.
- The NIH HEAL Initiative expects data sharing timelines to align with timeline requirements stated in the Final NIH Policy for Data Management and Sharing ( NOT-OD-21-013 ).
6. Additional Requirements for NIH HEAL Initiative studies conducting clinical research or research involving human subjects.
These studies must meet the following additional requirements:
- NIH HEAL Initiative trials that are required to register in clinicaltrials.gov should reference support from and inclusion in the NIH HEAL Initiative by including the standardized term “the HEAL Initiative ( https://heal.nih.gov/ )” in the Study Description Section.
- Studies that wish to use questionnaires not already included in the HEAL CDE repository should consult with their program official and the HEAL CDE team. New questionnaires will be considered for inclusion in the repository on a case-by-case basis and only when appropriate justification is provided.
- NIH HEAL Initiative clinical studies that are using copyrighted questionaries are required to obtain licenses for use prior to initiating data collection. Licenses must be shared with the HEAL CDE team and the program officer prior to use of copyrighted materials. For additional information, visit the HEAL CDE Program ( https://heal.nih.gov/data/common-data-elements ).
- To the extent possible, all other (nonpain) HEAL studies conducting clinical trials or research involving human subjects are expected to use questionnaires by the HEAL CDE Program ( https://heal.nih.gov/data/common-data-elements ) if applicable and relevant to their research.
Additional details, resources, and tools to assist with data-related activities can be found at https://www.healdatafair.org . Budgeting guidance for data sharing can be found in NOT-OD-21-015 and the NIH Scientific Data Sharing site .
All data collected as part of the NIH HEAL Initiative are collected under a Certificate of Confidentiality and entitled to the protections thereof. Institutions who receive data and/or materials from this award for performance of activities under this award are required to use the data and/or materials only as outlined by the NIH HEAL Initiative, in a manner that is consistent with applicable state and Federal laws and regulations, including any informed consent requirements and the terms of the institution’s NIH funding, including NOT-OD-17-109 and 42 U.S.C. 241(d). Failure to adhere to this criterion may result in enforcement actions.
4. Reporting
Progress reports for multi-year funded awards are due annually on or before the anniversary of the budget/project period start date of award. The reporting period for multi-year funded award progress report is the calendar year preceding the anniversary date of the award. Information on the content of the progress report and instructions on how to submit the report using the RPPR are posted at http://grants.nih.gov/grants/policy/myf.htm
- Recipients will provide updates at least annually on implementation of the PEDP.
( To follow the next section ):
Report and ensure immediate public access to HEAL-funded publications
Publications resulting from NIH HEAL Initiative-funded studies must be immediately publicly available upon publication.
- For manuscripts published in journals that are not immediately open access, authors should arrange with journals in advance to pay for immediate open access.
- Costs to ensure manuscripts are immediately publicly available upon publication should be included in budget requests.
Prior to publication, the NIH HEAL Initiative expects investigators to alert their program officers of upcoming manuscripts to ensure coordination of communication and outreach efforts.
Award recipients and their collaborators are required to acknowledge NIH HEAL Initiative support by referencing in the acknowledgment sections of any relevant publication:
“This research was supported by the National Institutes of Health through the NIH HEAL Initiative ( https://heal.nih.gov ) under award number [include specific grant/contract/award number; with NIH grant number(s) in this format: R01GM987654].”
A final RPPR, invention statement, and the expenditure data portion of the Federal Financial Report are required for closeout of an award, as described in the NIH Grants Policy Statement Section 8.6 Closeout . NIH NOFOs outline intended research goals and objectives. Post award, NIH will review and measure performance based on the details and outcomes that are shared within the RPPR, as described at 2 CFR Part 200.301.
Section VII. Agency Contacts
We encourage inquiries concerning this funding opportunity and welcome the opportunity to answer questions from potential applicants.
eRA Service Desk (Questions regarding ASSIST, eRA Commons, application errors and warnings, documenting system problems that threaten submission by the due date, and post-submission issues)
Finding Help Online: https://www.era.nih.gov/need-help (preferred method of contact) Telephone: 301-402-7469 or 866-504-9552 (Toll Free)
General Grants Information (Questions regarding application instructions, application processes, and NIH grant resources) Email: [email protected] (preferred method of contact) Telephone: 301-480-7075
Grants.gov Customer Support (Questions regarding Grants.gov registration and Workspace) Contact Center Telephone: 800-518-4726 Email: [email protected]
Alex Tuttle, Ph.D. National Center for Complementary and Integrative Health (NCCIH) Phone: 301-814-6115 Email: [email protected]
Mark Egli, Ph.D. National Institute on Alcohol Abuse and Alcoholism (NIAAA) Phone: 301-594-6382 E-mail: [email protected]
Rebecca N Lenzi, Ph.D. NATIONAL INSTITUTE OF ARTHRITIS AND MUSCULOSKELETAL AND SKIN DISEASES (NIAMS) Phone: (301) 402-2446 E-mail: [email protected]
Rene Etcheberrigaray, M.D. National Institute on Aging (NIA) Phone: 301-451-9798 Email: [email protected]
Susan Marden, PhD, RN Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) Telephone: 301-435-6838 Email: [email protected]
Elizabeth Sypek, PhD National Institute of Neurological Disorders and Stroke (NINDS) Email: [email protected]
Examine your eRA Commons account for review assignment and contact information (information appears 2 weeks after the submission due date).
Debbie Chen National Center for Complementary and Integrative Health (NCCIH) Phone: 301-594-3788 Email: [email protected]
Judy Fox National Institute on Alcohol Abuse and Alcoholism (NIAAA) Telephone: 301-443-4704 Email: [email protected]
Erik Edgerton NATIONAL INSTITUTE OF ARTHRITIS AND MUSCULOSKELETAL AND SKIN DISEASES (NIAMS) Phone: 301-594-7760 E-mail: [email protected]
Ryan Blakeney National Institute on Aging (NIA) Phone: 301-451-9802 Email: [email protected]
Margaret Young Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) Telephone: 301-642-4552 Email: [email protected]
Section VIII. Other Information
Recently issued trans-NIH policy notices may affect your application submission. A full list of policy notices published by NIH is provided in the NIH Guide for Grants and Contracts . All awards are subject to the terms and conditions, cost principles, and other considerations described in the NIH Grants Policy Statement .
Awards are made under the authorization of Sections 301 and 405 of the Public Health Service Act as amended (42 USC 241 and 284) and under Federal Regulations 42 CFR Part 52 and 2 CFR Part 200.


IMAGES
VIDEO
COMMENTS
Formulating research objectives has the following five steps, which could help researchers develop a clear objective: 8. Identify the research problem. Review past studies on subjects similar to your problem statement, that is, studies that use similar methods, variables, etc.
Example: Research objectives. To assess the relationship between sedentary habits and muscle atrophy among the participants. To determine the impact of dietary factors, particularly protein consumption, on the muscular health of the participants. To determine the effect of physical activity on the participants' muscular health.
Research questions, hypotheses and objectives. There is an increasing familiarity with the principles of evidence-based medicine in the surgical community. As surgeons become more aware of the hierarchy of evidence, grades of recommendations and the principles of critical appraisal, they develop an increasing familiarity with research design.
The research aims, objectives and research questions (collectively called the "golden thread") are arguably the most important thing you need to get right when you're crafting a research proposal, dissertation or thesis.We receive questions almost every day about this "holy trinity" of research and there's certainly a lot of confusion out there, so we've crafted this post to help ...
3. Simple hypothesis. A simple hypothesis is a statement made to reflect the relation between exactly two variables. One independent and one dependent. Consider the example, "Smoking is a prominent cause of lung cancer." The dependent variable, lung cancer, is dependent on the independent variable, smoking. 4.
INTRODUCTION. Scientific research is usually initiated by posing evidenced-based research questions which are then explicitly restated as hypotheses.1,2 The hypotheses provide directions to guide the study, solutions, explanations, and expected results.3,4 Both research questions and hypotheses are essentially formulated based on conventional theories and real-world processes, which allow the ...
and hypotheses, and sometimes objectives, to shape and specifically focus the purpose of the study. Quantitative research questionsinquire about the relationships among variables that the investigator seeks to know. They are used frequently in social science research and especially in survey studies. Quantitative hypotheses, on the other hand ...
2-sided hypotheses are generally preferred unless there's a strong justification for a 1-sided hypothesis. A solid research hypothesis, informed by a good research question, influences the research design and paves the way for defining clear research objectives. Types of Research Hypothesis. Y- and X-Centered Research Designs
The specific group being studied. The predicted outcome of the experiment or analysis. 5. Phrase your hypothesis in three ways. To identify the variables, you can write a simple prediction in if…then form. The first part of the sentence states the independent variable and the second part states the dependent variable.
A research objective is defined as a clear and concise statement of the specific goals and aims of a research study. It outlines what the researcher intends to accomplish and what they hope to learn or discover through their research. Research objectives are crucial for guiding the research process and ensuring that the study stays focused and ...
Research Objectives. Research objectives refer to the specific goals or aims of a research study. They provide a clear and concise description of what the researcher hopes to achieve by conducting the research.The objectives are typically based on the research questions and hypotheses formulated at the beginning of the study and are used to guide the research process.
The number of hypotheses should be determined based on the research objectives, research questions, and the complexity of the research topic. It is important to ensure that the hypotheses are focused, testable, and directly related to the research aims. 9. Can research hypotheses be used in qualitative research?
The first question asks for a ready-made solution, and is not focused or researchable. The second question is a clearer comparative question, but note that it may not be practically feasible. For a smaller research project or thesis, it could be narrowed down further to focus on the effectiveness of drunk driving laws in just one or two countries.
Additionally, research objectives are integral components of the research framework, establishing a clear direction that aligns with the overall research questions and hypotheses. This alignment helps to ensure that the study remains focused and relevant, facilitating the systematic collection, analysis, and interpretation of data.
Examples of research hypotheses can guide researchers in writing effective ones. In this blog, we'll learn what a research hypothesis is, why it's important in research, and the different types used in science. ... Choose research methods that align with your study objectives, such as experiments, surveys, or observational studies. The ...
A research hypothesis (or scientific hypothesis) is a statement about an expected relationship between variables, or explanation of an occurrence, that is clear, specific and testable. So, when you write up hypotheses for your dissertation or thesis, make sure that they meet all these criteria. If you do, you'll not only have rock-solid ...
Summary. One of the most important aspects of a thesis, dissertation or research paper is the correct formulation of the aims and objectives. This is because your aims and objectives will establish the scope, depth and direction that your research will ultimately take. An effective set of aims and objectives will give your research focus and ...
HYPOTHESES & RESEARCH QUESTIONS Nature of Hypothesis The hypothesis is a clear statement of what is intended to be investigated. It should be specified before research is conducted and openly stated in reporting the results. This allows to: Identify the research objectives Identify the key abstract concepts involved in the research
uestion, hypothesis and study objectives:Study: Warden SJ, Metcalf BR, Kiss ZS, et al. Low intensity pulsed ultrasound for chronic patellar tendinopathy: a randomi. d, double blind, placebo cont. olled trial. Rheumatology 2008;47:467-71.Research question: How does low intensity pulsed ultrasound (LIPUS) compare with a placebo device in ...
A good research question is essential to guide your research paper, dissertation, or thesis. All research questions should be: Focused on a single problem or issue. Researchable using primary and/or secondary sources. Feasible to answer within the timeframe and practical constraints. Specific enough to answer thoroughly.
The primary objective of a study is paired with the hypothesis of the study, and should be clearly stated in the introduction of the research protocol. Objectives usually state exactly the outcome measures that are going to be used within their statements. Strong verbs like determine, measure, assess, evaluate, identify, examine, investigate ...
The steps to write a research hypothesis are: 1. Stating the problem: Ensure that the hypothesis defines the research problem. 2. Writing a hypothesis as an 'if-then' statement: Include the action and the expected outcome of your study by following a 'if-then' structure.
In every research, the terms 'research aim', 'research objectives', 'research questions' and 'research hypotheses' tend to have precise meaning, therefore defining the core objectives is the ...
Objective 1: Develop Small-Scale Basic and Mechanistic Pain Research Projects. Proposed research projects should be hypothesis driven and use a rigorous scientific design to generate research data/evidence and advance scientific knowledge. Applications should include objectives that are attainable within the 3-year grant period.